User login
Nutrition, Drugs, or Bariatric Surgery: What’s the Best Approach for Sustained Weight Loss?
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).
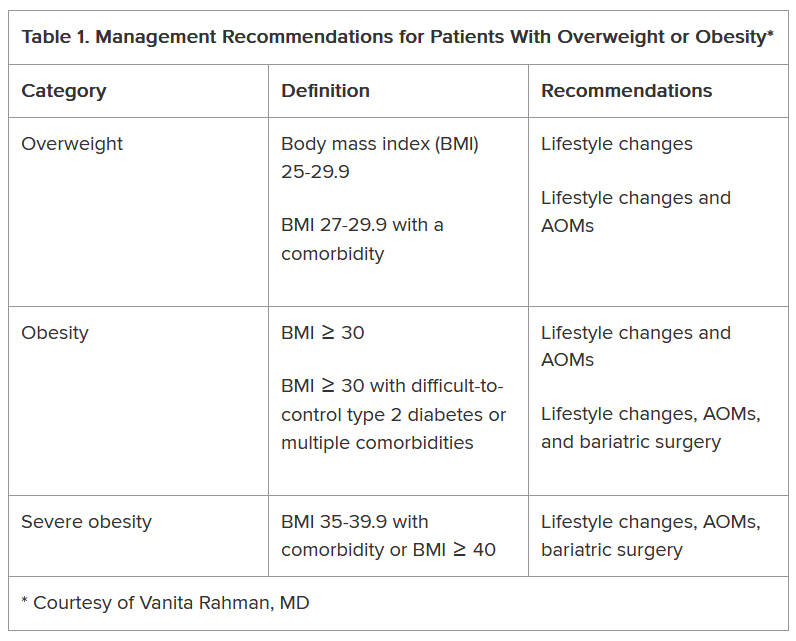
As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
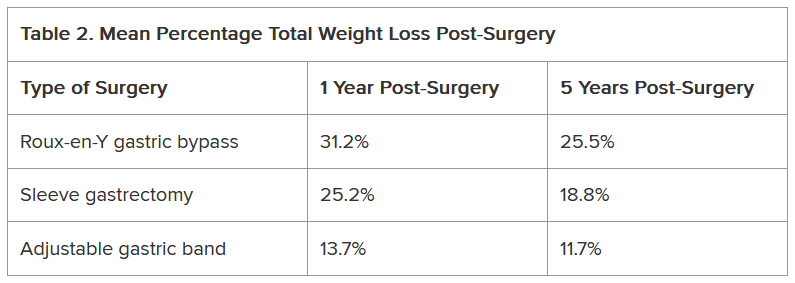
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).
Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).

As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).

Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).

As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).

Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
Are Patients On GLP-1s Getting the Right Nutrients?
As the use of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) continues to exponentially expand obesity treatment, concerns have arisen regarding their impact on nutrition in people who take them.
While the medications’ dampening effects on appetite result in an average weight reduction ≥ 15%, they also pose a risk for malnutrition.
“It’s important to eat a balanced diet when taking these medications,” Deena Adimoolam, MD, an endocrinologist based in New York City and a member of the national advisory committees for the Endocrine Society and the American Diabetes Association, said in an interview.
The decreased caloric intake resulting from the use of GLP-1 RAs makes it essential for patients to consume nutrient-dense foods. Clinicians can help patients achieve a healthy diet by anticipating nutrition problems, advising them on recommended target ranges of nutrient intake, and referring them for appropriate counseling.
Where to Begin
The task begins with “setting the right expectations before the patient starts treatment,” said Scott Isaacs, MD, president-elect of the American Association of Clinical Endocrinology.
To that end, it’s important to explain to patients how the medications affect appetite and how to adapt. GLP-1 RAs don’t completely turn off the appetite, and the effect at the beginning will likely be very mild, Isaacs said in an interview.
Some patients don’t notice a change for 2-3 months, although others see an effect sooner.
“Typically, people will notice that the main impact is on satiation, meaning they’ll fill up more quickly,” said Isaacs, who is an adjunct associate professor at Emory University School of Medicine, Atlanta, Georgia. “It’s important to tell them to stop eating when they feel full because eating when full can increase the side effects, such as nausea, vomiting, diarrhea, and constipation.”
A review article, written by lead author Jaime Almandoz, MD, University of Texas Southwestern Medical Center, Dallas, in Obesity offers a “5 A’s model” as a guide on how to begin discussing overweight or obesity with patients. This involves asking for permission to discuss weight and asking about food and vitamin/supplement intake; assessing the patient’s medical history and root causes of obesity, and conducting a physical examination; advising the patient regarding treatment options and reasonable expectations; agreeing on treatment and lifestyle goals; and assisting the patient to address challenges, referring them as needed to for additional support (eg, a dietitian), as well as arranging for follow-up.
Impact of GLP-1 RAs on Food Preferences
Besides reducing hunger and increasing satiety, GLP-1 RAs may affect food preferences, according to a research review published in The International Journal of Obesity. It cites a 2014 study that found that people taking GLP-1 RAs displayed decreased neuronal responses to images of food measured by functional magnetic resonance imaging in the areas of brain associated with appetite and reward. This might affect taste preferences and food intake.
Additionally, a 2023 study suggested that during the weight-loss phase of treatment (as opposed to the maintenance phase), patients may experience reduced cravings for dairy and starchy food, less desire to eat salty or spicy foods, and less difficulty controlling eating and resisting cravings.
“Altered food preferences, decreased food cravings, and reduced food intake may contribute to long-term weight loss,” according to the research review. Tailored treatments focusing on the weight maintenance phase are needed, the authors wrote.
Are Patients Vulnerable to Malnutrition?
A recent review found that total caloric intake was reduced by 16%-39% in patients taking a GLP-1 RA or dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA, but few studies evaluated the composition of these patients’ diets. Research that examines the qualitative changes in macronutrient and micronutrient intake of patients on these medications is needed, the authors wrote.
They outlined several nutritional concerns, including whether GLP-1 RA or GIP/ GLP-1 RA use could result in protein intake insufficient for maintaining muscle strength, mass, and function or in inadequate dietary quality (ie, poor intake of micronutrients, fiber, and fluid).
“Although we don’t necessarily see ‘malnutrition’ in our practice, we do see patients who lose too much weight after months and months of treatment, patients who aren’t hungry and don’t eat all day and have one big meal at the end of the day because they don’t feel like eating, and people who continue to eat unhealthy foods,” Isaacs said.
Some patients, however, have medical histories placing them at a greater risk for malnutrition. “Identification of these individuals may help prevent more serious nutritional and medical complications that might occur with decreased food intake associated with AOMs [anti-obesity medications],” Almandoz and colleagues noted in their review.
What Should Patients Eat?
Nutritional needs vary based on the patient’s age, sex, body weight, physical activity, and other factors, Almandoz and colleagues wrote. For this reason, energy intake during weight loss should be “personalized.”
The authors also recommended specific sources of the various dietary components and noted red flags signaling potential deficiencies
Nutritional needs vary based on the degree of appetite suppression in the patient, Adimoolam said. “I recommend at least two servings of fruits and vegetables daily, and drinking plenty of water throughout the day,” she added.
Protein in particular is a “key macronutrient,” and insufficient intake can lead to a variety of adverse effects, including sarcopenia — which is already a concern in individuals being treated with GLP-1 RAs. Meal replacement products (eg, shakes or bars) can supplement diets to help meet protein needs, especially if appetite is significantly reduced.
“There are definitely concerns for sarcopenia, so we have our patients taking these drugs try to eat healthy lean proteins – 100 g/d — and exercise,” Isaacs said. Exercise, including resistance training, not only improves muscle mass but also potentiates the effects of the GLP-1 RAs in patients with obesity and type 2 diabetes.
Adequate hydration is essential for patients taking GLP-1 RAs. “One of the commonly described side effects is fatigue, but there’s no biological reason why these medications should cause fatigue. My opinion is that these patients are dehydrated, and that may be causing the fatigue,” Isaacs said.
Some patients taking GLP-1 RAs lose interest in food. Isaacs regarded this as an “adverse reaction to the medication, which necessitates either stopping it altogether, changing the dose, or adjusting the diet.” There are “many different solutions, and one size doesn’t fit all,” he said.
Dietary and Behavioral Counseling
The drugs don’t necessarily motivate a person to eat healthier food, only to eat less food, Isaacs noted.
“The person might be eating low-volume but high-calorie food, such as bag of chips or a cookie instead of an apple,” Isaacs said. Patients who are losing weight “may not realize that weight loss isn’t the only important outcome. Because they’re losing weight, they think it’s okay to eat junk food.”
Patients need education and guidance about how to eat while on these medications. Most patients find counseling about meal planning helpful, he said.
Isaacs gives nutritional guidance to his patients when he prescribes a weight loss medication. “But most physicians don’t have time to offer that type of specific counseling on an ongoing basis,” he said. Isaacs refers patients requiring more detailed and long-term guidance to a dietitian.
Patients with monotonous diets of poor quality are at increased risk for nutrition deficiencies, and counseling by a registered dietitian could help improve their dietary quality.
Registered dietitians can develop a multifaceted approach not only focusing on medication management but also on customizing the patient’s diet, assisting with lifestyle adjustments, and addressing the mental health issues surrounding obesity and its management.
People seeking obesity treatment often have psychiatric conditions, psychological distress, or disordered eating patterns, and questions and concerns have emerged about how GLP-1 RA use might affect existing mental health problems. For example, if the medication suppresses the feeling of gratification a person once got from eating high-energy dense foods, that individual may “seek rewards or pleasure elsewhere, and possibly from unhealthy sources.”
Psychological issues also may emerge as a result of weight loss, so it’s helpful to take a multidisciplinary approach that includes mental health practitioners to support patients who are being treated with GLP-1 RAs. Patients taking these agents should be monitored for the emergence or worsening of psychiatric conditions, such as depression and suicidal ideation.
Achieving significant weight loss may lead to “unexpected changes” in the dynamics of patients’ relationship with others, “which can be distressing.” Clinicians should be “sensitive to patients’ social and emotional needs” and provide support or refer patients for help with coping strategies.
GLP-1 RAs have enormous potential to improve health outcomes in patients with obesity. Careful patient selection, close monitoring, and support for patients with nutrition and other lifestyle issues can increase the chances that these agents will fulfill their potential.
Isaacs declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
As the use of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) continues to exponentially expand obesity treatment, concerns have arisen regarding their impact on nutrition in people who take them.
While the medications’ dampening effects on appetite result in an average weight reduction ≥ 15%, they also pose a risk for malnutrition.
“It’s important to eat a balanced diet when taking these medications,” Deena Adimoolam, MD, an endocrinologist based in New York City and a member of the national advisory committees for the Endocrine Society and the American Diabetes Association, said in an interview.
The decreased caloric intake resulting from the use of GLP-1 RAs makes it essential for patients to consume nutrient-dense foods. Clinicians can help patients achieve a healthy diet by anticipating nutrition problems, advising them on recommended target ranges of nutrient intake, and referring them for appropriate counseling.
Where to Begin
The task begins with “setting the right expectations before the patient starts treatment,” said Scott Isaacs, MD, president-elect of the American Association of Clinical Endocrinology.
To that end, it’s important to explain to patients how the medications affect appetite and how to adapt. GLP-1 RAs don’t completely turn off the appetite, and the effect at the beginning will likely be very mild, Isaacs said in an interview.
Some patients don’t notice a change for 2-3 months, although others see an effect sooner.
“Typically, people will notice that the main impact is on satiation, meaning they’ll fill up more quickly,” said Isaacs, who is an adjunct associate professor at Emory University School of Medicine, Atlanta, Georgia. “It’s important to tell them to stop eating when they feel full because eating when full can increase the side effects, such as nausea, vomiting, diarrhea, and constipation.”
A review article, written by lead author Jaime Almandoz, MD, University of Texas Southwestern Medical Center, Dallas, in Obesity offers a “5 A’s model” as a guide on how to begin discussing overweight or obesity with patients. This involves asking for permission to discuss weight and asking about food and vitamin/supplement intake; assessing the patient’s medical history and root causes of obesity, and conducting a physical examination; advising the patient regarding treatment options and reasonable expectations; agreeing on treatment and lifestyle goals; and assisting the patient to address challenges, referring them as needed to for additional support (eg, a dietitian), as well as arranging for follow-up.
Impact of GLP-1 RAs on Food Preferences
Besides reducing hunger and increasing satiety, GLP-1 RAs may affect food preferences, according to a research review published in The International Journal of Obesity. It cites a 2014 study that found that people taking GLP-1 RAs displayed decreased neuronal responses to images of food measured by functional magnetic resonance imaging in the areas of brain associated with appetite and reward. This might affect taste preferences and food intake.
Additionally, a 2023 study suggested that during the weight-loss phase of treatment (as opposed to the maintenance phase), patients may experience reduced cravings for dairy and starchy food, less desire to eat salty or spicy foods, and less difficulty controlling eating and resisting cravings.
“Altered food preferences, decreased food cravings, and reduced food intake may contribute to long-term weight loss,” according to the research review. Tailored treatments focusing on the weight maintenance phase are needed, the authors wrote.
Are Patients Vulnerable to Malnutrition?
A recent review found that total caloric intake was reduced by 16%-39% in patients taking a GLP-1 RA or dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA, but few studies evaluated the composition of these patients’ diets. Research that examines the qualitative changes in macronutrient and micronutrient intake of patients on these medications is needed, the authors wrote.
They outlined several nutritional concerns, including whether GLP-1 RA or GIP/ GLP-1 RA use could result in protein intake insufficient for maintaining muscle strength, mass, and function or in inadequate dietary quality (ie, poor intake of micronutrients, fiber, and fluid).
“Although we don’t necessarily see ‘malnutrition’ in our practice, we do see patients who lose too much weight after months and months of treatment, patients who aren’t hungry and don’t eat all day and have one big meal at the end of the day because they don’t feel like eating, and people who continue to eat unhealthy foods,” Isaacs said.
Some patients, however, have medical histories placing them at a greater risk for malnutrition. “Identification of these individuals may help prevent more serious nutritional and medical complications that might occur with decreased food intake associated with AOMs [anti-obesity medications],” Almandoz and colleagues noted in their review.
What Should Patients Eat?
Nutritional needs vary based on the patient’s age, sex, body weight, physical activity, and other factors, Almandoz and colleagues wrote. For this reason, energy intake during weight loss should be “personalized.”
The authors also recommended specific sources of the various dietary components and noted red flags signaling potential deficiencies
Nutritional needs vary based on the degree of appetite suppression in the patient, Adimoolam said. “I recommend at least two servings of fruits and vegetables daily, and drinking plenty of water throughout the day,” she added.
Protein in particular is a “key macronutrient,” and insufficient intake can lead to a variety of adverse effects, including sarcopenia — which is already a concern in individuals being treated with GLP-1 RAs. Meal replacement products (eg, shakes or bars) can supplement diets to help meet protein needs, especially if appetite is significantly reduced.
“There are definitely concerns for sarcopenia, so we have our patients taking these drugs try to eat healthy lean proteins – 100 g/d — and exercise,” Isaacs said. Exercise, including resistance training, not only improves muscle mass but also potentiates the effects of the GLP-1 RAs in patients with obesity and type 2 diabetes.
Adequate hydration is essential for patients taking GLP-1 RAs. “One of the commonly described side effects is fatigue, but there’s no biological reason why these medications should cause fatigue. My opinion is that these patients are dehydrated, and that may be causing the fatigue,” Isaacs said.
Some patients taking GLP-1 RAs lose interest in food. Isaacs regarded this as an “adverse reaction to the medication, which necessitates either stopping it altogether, changing the dose, or adjusting the diet.” There are “many different solutions, and one size doesn’t fit all,” he said.
Dietary and Behavioral Counseling
The drugs don’t necessarily motivate a person to eat healthier food, only to eat less food, Isaacs noted.
“The person might be eating low-volume but high-calorie food, such as bag of chips or a cookie instead of an apple,” Isaacs said. Patients who are losing weight “may not realize that weight loss isn’t the only important outcome. Because they’re losing weight, they think it’s okay to eat junk food.”
Patients need education and guidance about how to eat while on these medications. Most patients find counseling about meal planning helpful, he said.
Isaacs gives nutritional guidance to his patients when he prescribes a weight loss medication. “But most physicians don’t have time to offer that type of specific counseling on an ongoing basis,” he said. Isaacs refers patients requiring more detailed and long-term guidance to a dietitian.
Patients with monotonous diets of poor quality are at increased risk for nutrition deficiencies, and counseling by a registered dietitian could help improve their dietary quality.
Registered dietitians can develop a multifaceted approach not only focusing on medication management but also on customizing the patient’s diet, assisting with lifestyle adjustments, and addressing the mental health issues surrounding obesity and its management.
People seeking obesity treatment often have psychiatric conditions, psychological distress, or disordered eating patterns, and questions and concerns have emerged about how GLP-1 RA use might affect existing mental health problems. For example, if the medication suppresses the feeling of gratification a person once got from eating high-energy dense foods, that individual may “seek rewards or pleasure elsewhere, and possibly from unhealthy sources.”
Psychological issues also may emerge as a result of weight loss, so it’s helpful to take a multidisciplinary approach that includes mental health practitioners to support patients who are being treated with GLP-1 RAs. Patients taking these agents should be monitored for the emergence or worsening of psychiatric conditions, such as depression and suicidal ideation.
Achieving significant weight loss may lead to “unexpected changes” in the dynamics of patients’ relationship with others, “which can be distressing.” Clinicians should be “sensitive to patients’ social and emotional needs” and provide support or refer patients for help with coping strategies.
GLP-1 RAs have enormous potential to improve health outcomes in patients with obesity. Careful patient selection, close monitoring, and support for patients with nutrition and other lifestyle issues can increase the chances that these agents will fulfill their potential.
Isaacs declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
As the use of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) continues to exponentially expand obesity treatment, concerns have arisen regarding their impact on nutrition in people who take them.
While the medications’ dampening effects on appetite result in an average weight reduction ≥ 15%, they also pose a risk for malnutrition.
“It’s important to eat a balanced diet when taking these medications,” Deena Adimoolam, MD, an endocrinologist based in New York City and a member of the national advisory committees for the Endocrine Society and the American Diabetes Association, said in an interview.
The decreased caloric intake resulting from the use of GLP-1 RAs makes it essential for patients to consume nutrient-dense foods. Clinicians can help patients achieve a healthy diet by anticipating nutrition problems, advising them on recommended target ranges of nutrient intake, and referring them for appropriate counseling.
Where to Begin
The task begins with “setting the right expectations before the patient starts treatment,” said Scott Isaacs, MD, president-elect of the American Association of Clinical Endocrinology.
To that end, it’s important to explain to patients how the medications affect appetite and how to adapt. GLP-1 RAs don’t completely turn off the appetite, and the effect at the beginning will likely be very mild, Isaacs said in an interview.
Some patients don’t notice a change for 2-3 months, although others see an effect sooner.
“Typically, people will notice that the main impact is on satiation, meaning they’ll fill up more quickly,” said Isaacs, who is an adjunct associate professor at Emory University School of Medicine, Atlanta, Georgia. “It’s important to tell them to stop eating when they feel full because eating when full can increase the side effects, such as nausea, vomiting, diarrhea, and constipation.”
A review article, written by lead author Jaime Almandoz, MD, University of Texas Southwestern Medical Center, Dallas, in Obesity offers a “5 A’s model” as a guide on how to begin discussing overweight or obesity with patients. This involves asking for permission to discuss weight and asking about food and vitamin/supplement intake; assessing the patient’s medical history and root causes of obesity, and conducting a physical examination; advising the patient regarding treatment options and reasonable expectations; agreeing on treatment and lifestyle goals; and assisting the patient to address challenges, referring them as needed to for additional support (eg, a dietitian), as well as arranging for follow-up.
Impact of GLP-1 RAs on Food Preferences
Besides reducing hunger and increasing satiety, GLP-1 RAs may affect food preferences, according to a research review published in The International Journal of Obesity. It cites a 2014 study that found that people taking GLP-1 RAs displayed decreased neuronal responses to images of food measured by functional magnetic resonance imaging in the areas of brain associated with appetite and reward. This might affect taste preferences and food intake.
Additionally, a 2023 study suggested that during the weight-loss phase of treatment (as opposed to the maintenance phase), patients may experience reduced cravings for dairy and starchy food, less desire to eat salty or spicy foods, and less difficulty controlling eating and resisting cravings.
“Altered food preferences, decreased food cravings, and reduced food intake may contribute to long-term weight loss,” according to the research review. Tailored treatments focusing on the weight maintenance phase are needed, the authors wrote.
Are Patients Vulnerable to Malnutrition?
A recent review found that total caloric intake was reduced by 16%-39% in patients taking a GLP-1 RA or dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA, but few studies evaluated the composition of these patients’ diets. Research that examines the qualitative changes in macronutrient and micronutrient intake of patients on these medications is needed, the authors wrote.
They outlined several nutritional concerns, including whether GLP-1 RA or GIP/ GLP-1 RA use could result in protein intake insufficient for maintaining muscle strength, mass, and function or in inadequate dietary quality (ie, poor intake of micronutrients, fiber, and fluid).
“Although we don’t necessarily see ‘malnutrition’ in our practice, we do see patients who lose too much weight after months and months of treatment, patients who aren’t hungry and don’t eat all day and have one big meal at the end of the day because they don’t feel like eating, and people who continue to eat unhealthy foods,” Isaacs said.
Some patients, however, have medical histories placing them at a greater risk for malnutrition. “Identification of these individuals may help prevent more serious nutritional and medical complications that might occur with decreased food intake associated with AOMs [anti-obesity medications],” Almandoz and colleagues noted in their review.
What Should Patients Eat?
Nutritional needs vary based on the patient’s age, sex, body weight, physical activity, and other factors, Almandoz and colleagues wrote. For this reason, energy intake during weight loss should be “personalized.”
The authors also recommended specific sources of the various dietary components and noted red flags signaling potential deficiencies
Nutritional needs vary based on the degree of appetite suppression in the patient, Adimoolam said. “I recommend at least two servings of fruits and vegetables daily, and drinking plenty of water throughout the day,” she added.
Protein in particular is a “key macronutrient,” and insufficient intake can lead to a variety of adverse effects, including sarcopenia — which is already a concern in individuals being treated with GLP-1 RAs. Meal replacement products (eg, shakes or bars) can supplement diets to help meet protein needs, especially if appetite is significantly reduced.
“There are definitely concerns for sarcopenia, so we have our patients taking these drugs try to eat healthy lean proteins – 100 g/d — and exercise,” Isaacs said. Exercise, including resistance training, not only improves muscle mass but also potentiates the effects of the GLP-1 RAs in patients with obesity and type 2 diabetes.
Adequate hydration is essential for patients taking GLP-1 RAs. “One of the commonly described side effects is fatigue, but there’s no biological reason why these medications should cause fatigue. My opinion is that these patients are dehydrated, and that may be causing the fatigue,” Isaacs said.
Some patients taking GLP-1 RAs lose interest in food. Isaacs regarded this as an “adverse reaction to the medication, which necessitates either stopping it altogether, changing the dose, or adjusting the diet.” There are “many different solutions, and one size doesn’t fit all,” he said.
Dietary and Behavioral Counseling
The drugs don’t necessarily motivate a person to eat healthier food, only to eat less food, Isaacs noted.
“The person might be eating low-volume but high-calorie food, such as bag of chips or a cookie instead of an apple,” Isaacs said. Patients who are losing weight “may not realize that weight loss isn’t the only important outcome. Because they’re losing weight, they think it’s okay to eat junk food.”
Patients need education and guidance about how to eat while on these medications. Most patients find counseling about meal planning helpful, he said.
Isaacs gives nutritional guidance to his patients when he prescribes a weight loss medication. “But most physicians don’t have time to offer that type of specific counseling on an ongoing basis,” he said. Isaacs refers patients requiring more detailed and long-term guidance to a dietitian.
Patients with monotonous diets of poor quality are at increased risk for nutrition deficiencies, and counseling by a registered dietitian could help improve their dietary quality.
Registered dietitians can develop a multifaceted approach not only focusing on medication management but also on customizing the patient’s diet, assisting with lifestyle adjustments, and addressing the mental health issues surrounding obesity and its management.
People seeking obesity treatment often have psychiatric conditions, psychological distress, or disordered eating patterns, and questions and concerns have emerged about how GLP-1 RA use might affect existing mental health problems. For example, if the medication suppresses the feeling of gratification a person once got from eating high-energy dense foods, that individual may “seek rewards or pleasure elsewhere, and possibly from unhealthy sources.”
Psychological issues also may emerge as a result of weight loss, so it’s helpful to take a multidisciplinary approach that includes mental health practitioners to support patients who are being treated with GLP-1 RAs. Patients taking these agents should be monitored for the emergence or worsening of psychiatric conditions, such as depression and suicidal ideation.
Achieving significant weight loss may lead to “unexpected changes” in the dynamics of patients’ relationship with others, “which can be distressing.” Clinicians should be “sensitive to patients’ social and emotional needs” and provide support or refer patients for help with coping strategies.
GLP-1 RAs have enormous potential to improve health outcomes in patients with obesity. Careful patient selection, close monitoring, and support for patients with nutrition and other lifestyle issues can increase the chances that these agents will fulfill their potential.
Isaacs declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
Can GLP-1s Reduce Alzheimer’s Disease Risk?
Tina is a lovely 67-year-old woman who was recently found to be an APOE gene carrier (a gene associated with increased risk of developing Alzheimer’s disease as well as an earlier age of disease onset), with diffused amyloid protein deposition her brain.
Her neuropsychiatric testing was consistent with mild cognitive impairment. Although Tina is not a doctor herself, her entire family consists of doctors, and she came to me under their advisement to consider semaglutide (Ozempic) for early Alzheimer’s disease prevention.
This would usually be simple, but in Tina’s case, there was a complicating factor: At 5’ and 90 pounds, she was already considerably underweight and was at risk of becoming severely undernourished.
To understand the potential role for glucagon-like peptide-1 (GLP-1) receptor agonists such as Ozempic in prevention, a quick primer on Alzheimer’s Disease is necessary.
The exact cause of Alzheimer’s disease remains elusive, but it is probably due to a combination of factors, including:
- Buildup of abnormal amyloid and tau proteins around brain cells
- Brain shrinkage, with subsequent damage to blood vessels and mitochondria, and inflammation
- Genetic predisposition
- Lifestyle factors, including obesity, high blood pressure, high cholesterol, and diabetes.
Once in the brain, they can reduce inflammation and improve functioning of the neurons. In early rodent trials, GLP-1 receptor agonists led to reduced amyloid and tau aggregation, downregulation of inflammation, and improved memory.
In 2021, multiple studies showed that liraglutide, an early GLP-1 receptor agonist, improved cognitive function and MRI volume in patients with Alzheimer’s disease.
A study recently published in Alzheimer’s & Dementia analyzed data from 1 million people with type 2 diabetes and no prior Alzheimer’s disease diagnosis. The authors compared Alzheimer’s disease occurrence in patients taking various diabetes medications, including insulin, metformin, and GLP-1 receptor agonists. The study found that participants taking semaglutide had up to a 70% reduction in Alzheimer’s risk. The results were consistent across gender, age, and weight.
Given the reassuring safety profile of GLP-1 receptor agonists and lack of other effective treatment or prophylaxis for Alzheimer’s disease, I agreed to start her on dulaglutide (Trulicity). My rationale was twofold:
1. In studies, dulaglutide has the highest uptake in the brain tissue at 68%. By contrast, there is virtually zero uptake in brain tissue for semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound). Because this class of drugs exert their effects in the brain tissue, I wanted to give her a GLP-1 receptor agonist with a high percent uptake.
2. Trulicity has a minimal effect on weight loss compared with the newer-generation GLP-1 receptor agonists. Even so, I connected Tina to my dietitian to ensure that she would receive a high-protein, high-calorie diet.
Tina has now been taking Trulicity for 6 months. Although it is certainly too early to draw firm conclusions about the efficacy of her treatment, she is not experiencing any weight loss and is cognitively stable, according to her neurologist.
The EVOKE and EVOKE+ phase 3 trials are currently underway to evaluate the efficacy of semaglutide to treat mild cognitive impairment and early Alzheimer’s in amyloid-positive patients. Results are expected in 2025, but in the meantime, I feel comforted knowing that Tina is receiving a potentially beneficial and definitively low-risk treatment.
Dr Messer, Clinical Assistant Professor, Mount Sinai School of Medicine; Associate Professor, Hofstra School of Medicine, New York, NY, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Tina is a lovely 67-year-old woman who was recently found to be an APOE gene carrier (a gene associated with increased risk of developing Alzheimer’s disease as well as an earlier age of disease onset), with diffused amyloid protein deposition her brain.
Her neuropsychiatric testing was consistent with mild cognitive impairment. Although Tina is not a doctor herself, her entire family consists of doctors, and she came to me under their advisement to consider semaglutide (Ozempic) for early Alzheimer’s disease prevention.
This would usually be simple, but in Tina’s case, there was a complicating factor: At 5’ and 90 pounds, she was already considerably underweight and was at risk of becoming severely undernourished.
To understand the potential role for glucagon-like peptide-1 (GLP-1) receptor agonists such as Ozempic in prevention, a quick primer on Alzheimer’s Disease is necessary.
The exact cause of Alzheimer’s disease remains elusive, but it is probably due to a combination of factors, including:
- Buildup of abnormal amyloid and tau proteins around brain cells
- Brain shrinkage, with subsequent damage to blood vessels and mitochondria, and inflammation
- Genetic predisposition
- Lifestyle factors, including obesity, high blood pressure, high cholesterol, and diabetes.
Once in the brain, they can reduce inflammation and improve functioning of the neurons. In early rodent trials, GLP-1 receptor agonists led to reduced amyloid and tau aggregation, downregulation of inflammation, and improved memory.
In 2021, multiple studies showed that liraglutide, an early GLP-1 receptor agonist, improved cognitive function and MRI volume in patients with Alzheimer’s disease.
A study recently published in Alzheimer’s & Dementia analyzed data from 1 million people with type 2 diabetes and no prior Alzheimer’s disease diagnosis. The authors compared Alzheimer’s disease occurrence in patients taking various diabetes medications, including insulin, metformin, and GLP-1 receptor agonists. The study found that participants taking semaglutide had up to a 70% reduction in Alzheimer’s risk. The results were consistent across gender, age, and weight.
Given the reassuring safety profile of GLP-1 receptor agonists and lack of other effective treatment or prophylaxis for Alzheimer’s disease, I agreed to start her on dulaglutide (Trulicity). My rationale was twofold:
1. In studies, dulaglutide has the highest uptake in the brain tissue at 68%. By contrast, there is virtually zero uptake in brain tissue for semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound). Because this class of drugs exert their effects in the brain tissue, I wanted to give her a GLP-1 receptor agonist with a high percent uptake.
2. Trulicity has a minimal effect on weight loss compared with the newer-generation GLP-1 receptor agonists. Even so, I connected Tina to my dietitian to ensure that she would receive a high-protein, high-calorie diet.
Tina has now been taking Trulicity for 6 months. Although it is certainly too early to draw firm conclusions about the efficacy of her treatment, she is not experiencing any weight loss and is cognitively stable, according to her neurologist.
The EVOKE and EVOKE+ phase 3 trials are currently underway to evaluate the efficacy of semaglutide to treat mild cognitive impairment and early Alzheimer’s in amyloid-positive patients. Results are expected in 2025, but in the meantime, I feel comforted knowing that Tina is receiving a potentially beneficial and definitively low-risk treatment.
Dr Messer, Clinical Assistant Professor, Mount Sinai School of Medicine; Associate Professor, Hofstra School of Medicine, New York, NY, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Tina is a lovely 67-year-old woman who was recently found to be an APOE gene carrier (a gene associated with increased risk of developing Alzheimer’s disease as well as an earlier age of disease onset), with diffused amyloid protein deposition her brain.
Her neuropsychiatric testing was consistent with mild cognitive impairment. Although Tina is not a doctor herself, her entire family consists of doctors, and she came to me under their advisement to consider semaglutide (Ozempic) for early Alzheimer’s disease prevention.
This would usually be simple, but in Tina’s case, there was a complicating factor: At 5’ and 90 pounds, she was already considerably underweight and was at risk of becoming severely undernourished.
To understand the potential role for glucagon-like peptide-1 (GLP-1) receptor agonists such as Ozempic in prevention, a quick primer on Alzheimer’s Disease is necessary.
The exact cause of Alzheimer’s disease remains elusive, but it is probably due to a combination of factors, including:
- Buildup of abnormal amyloid and tau proteins around brain cells
- Brain shrinkage, with subsequent damage to blood vessels and mitochondria, and inflammation
- Genetic predisposition
- Lifestyle factors, including obesity, high blood pressure, high cholesterol, and diabetes.
Once in the brain, they can reduce inflammation and improve functioning of the neurons. In early rodent trials, GLP-1 receptor agonists led to reduced amyloid and tau aggregation, downregulation of inflammation, and improved memory.
In 2021, multiple studies showed that liraglutide, an early GLP-1 receptor agonist, improved cognitive function and MRI volume in patients with Alzheimer’s disease.
A study recently published in Alzheimer’s & Dementia analyzed data from 1 million people with type 2 diabetes and no prior Alzheimer’s disease diagnosis. The authors compared Alzheimer’s disease occurrence in patients taking various diabetes medications, including insulin, metformin, and GLP-1 receptor agonists. The study found that participants taking semaglutide had up to a 70% reduction in Alzheimer’s risk. The results were consistent across gender, age, and weight.
Given the reassuring safety profile of GLP-1 receptor agonists and lack of other effective treatment or prophylaxis for Alzheimer’s disease, I agreed to start her on dulaglutide (Trulicity). My rationale was twofold:
1. In studies, dulaglutide has the highest uptake in the brain tissue at 68%. By contrast, there is virtually zero uptake in brain tissue for semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound). Because this class of drugs exert their effects in the brain tissue, I wanted to give her a GLP-1 receptor agonist with a high percent uptake.
2. Trulicity has a minimal effect on weight loss compared with the newer-generation GLP-1 receptor agonists. Even so, I connected Tina to my dietitian to ensure that she would receive a high-protein, high-calorie diet.
Tina has now been taking Trulicity for 6 months. Although it is certainly too early to draw firm conclusions about the efficacy of her treatment, she is not experiencing any weight loss and is cognitively stable, according to her neurologist.
The EVOKE and EVOKE+ phase 3 trials are currently underway to evaluate the efficacy of semaglutide to treat mild cognitive impairment and early Alzheimer’s in amyloid-positive patients. Results are expected in 2025, but in the meantime, I feel comforted knowing that Tina is receiving a potentially beneficial and definitively low-risk treatment.
Dr Messer, Clinical Assistant Professor, Mount Sinai School of Medicine; Associate Professor, Hofstra School of Medicine, New York, NY, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
How Does End of Life Impact Diabetes Care?
TOPLINE:
Among older adults with type 2 diabetes (T2D), the use of antidiabetes medications declined in the last year before death, with notable shifts from metformin and sulfonylureas toward insulin therapy.
METHODOLOGY:
Current recommendations emphasize a more liberal approach to glycemic control in people with a high burden of comorbidities and shorter life expectancy, but little is known about the changes and discontinuation patterns of diabetes medications among older adults near the end of life.
.
All medication classes available during the study period were considered, including short-acting and long-acting insulins, metformin, sulfonylureas, dipeptidyl peptidase-4 inhibitors, glucagon-like peptide 1 (GLP-1) receptor agonists, sodium-glucose cotransporter-2 (SGLT2) inhibitors, and other medications.
Analysis included temporal trends in prescribing antidiabetes medications, stratified by frailty using a validated claims-based frailty index, with scores ≥ 0.30 indicating higher frailty.
Antidiabetes medication fills were assessed within 1 year before death, examining changes across three time periods: 12 to 8 months, 8 to 4 months, and 4 to 0 months before death.
TAKEAWAY:
The proportion of older patients receiving antidiabetes medications increased slightly from 71.4% in 2015 to 72.9% in 2019, with metformin showing the largest increase from 40.7% to 46.5% (standardized mean difference [SMD], −0.12) and sulfonylureas showing the largest decrease from 37.0% to 31.8% (SMD, 0.11).
The use of newer diabetes medications with cardiovascular benefits, such as GLP-1 receptor agonists and SGLT2 inhibitors, remained less common but showed increasing trends over time.
The use of any antidiabetes medication decreased from 66.1% in the 9 to 12 months before death to 60.8% in the last 4 months of life (P < .01), primarily due to the reduced use of metformin and sulfonylureas.
The use of both short-acting and long-acting insulin agents increased toward the end of life (from 28.0% to 32.9% and from 41.2% to 43.9%, respectively; both P < .001) , particularly among frailer individuals.
IN PRACTICE:
“[The study] findings underscore important implications for diabetes management in patients nearing the end of life. With ~70% of patients with T2D using at least one antidiabetes medication, there is a need to consider further de-escalation or deprescribing in this vulnerable population,” the authors wrote.
SOURCE:
The study was led by Alexander Kutz, MD, MPH, MSc, Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, and was published online in Diabetes Care.
LIMITATIONS:
The study lacked details on the reasons for changes in medication patterns, making it unclear whether these changes were due to clinical guidelines or to reduce adverse events. Moreover, the study could not capture transitions or substitutions between medications, information on the dosage data, and causes of death.
DISCLOSURES:
This study was supported by the Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School. Some authors reported receiving personal fees or research grants from the National Institutes of Health and other institutions and a few pharmaceutical companies. One author reported acting as a principal investigator and receiving a research grant from Boehringer-Ingelheim, unrelated to the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Among older adults with type 2 diabetes (T2D), the use of antidiabetes medications declined in the last year before death, with notable shifts from metformin and sulfonylureas toward insulin therapy.
METHODOLOGY:
Current recommendations emphasize a more liberal approach to glycemic control in people with a high burden of comorbidities and shorter life expectancy, but little is known about the changes and discontinuation patterns of diabetes medications among older adults near the end of life.
.
All medication classes available during the study period were considered, including short-acting and long-acting insulins, metformin, sulfonylureas, dipeptidyl peptidase-4 inhibitors, glucagon-like peptide 1 (GLP-1) receptor agonists, sodium-glucose cotransporter-2 (SGLT2) inhibitors, and other medications.
Analysis included temporal trends in prescribing antidiabetes medications, stratified by frailty using a validated claims-based frailty index, with scores ≥ 0.30 indicating higher frailty.
Antidiabetes medication fills were assessed within 1 year before death, examining changes across three time periods: 12 to 8 months, 8 to 4 months, and 4 to 0 months before death.
TAKEAWAY:
The proportion of older patients receiving antidiabetes medications increased slightly from 71.4% in 2015 to 72.9% in 2019, with metformin showing the largest increase from 40.7% to 46.5% (standardized mean difference [SMD], −0.12) and sulfonylureas showing the largest decrease from 37.0% to 31.8% (SMD, 0.11).
The use of newer diabetes medications with cardiovascular benefits, such as GLP-1 receptor agonists and SGLT2 inhibitors, remained less common but showed increasing trends over time.
The use of any antidiabetes medication decreased from 66.1% in the 9 to 12 months before death to 60.8% in the last 4 months of life (P < .01), primarily due to the reduced use of metformin and sulfonylureas.
The use of both short-acting and long-acting insulin agents increased toward the end of life (from 28.0% to 32.9% and from 41.2% to 43.9%, respectively; both P < .001) , particularly among frailer individuals.
IN PRACTICE:
“[The study] findings underscore important implications for diabetes management in patients nearing the end of life. With ~70% of patients with T2D using at least one antidiabetes medication, there is a need to consider further de-escalation or deprescribing in this vulnerable population,” the authors wrote.
SOURCE:
The study was led by Alexander Kutz, MD, MPH, MSc, Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, and was published online in Diabetes Care.
LIMITATIONS:
The study lacked details on the reasons for changes in medication patterns, making it unclear whether these changes were due to clinical guidelines or to reduce adverse events. Moreover, the study could not capture transitions or substitutions between medications, information on the dosage data, and causes of death.
DISCLOSURES:
This study was supported by the Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School. Some authors reported receiving personal fees or research grants from the National Institutes of Health and other institutions and a few pharmaceutical companies. One author reported acting as a principal investigator and receiving a research grant from Boehringer-Ingelheim, unrelated to the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Among older adults with type 2 diabetes (T2D), the use of antidiabetes medications declined in the last year before death, with notable shifts from metformin and sulfonylureas toward insulin therapy.
METHODOLOGY:
Current recommendations emphasize a more liberal approach to glycemic control in people with a high burden of comorbidities and shorter life expectancy, but little is known about the changes and discontinuation patterns of diabetes medications among older adults near the end of life.
.
All medication classes available during the study period were considered, including short-acting and long-acting insulins, metformin, sulfonylureas, dipeptidyl peptidase-4 inhibitors, glucagon-like peptide 1 (GLP-1) receptor agonists, sodium-glucose cotransporter-2 (SGLT2) inhibitors, and other medications.
Analysis included temporal trends in prescribing antidiabetes medications, stratified by frailty using a validated claims-based frailty index, with scores ≥ 0.30 indicating higher frailty.
Antidiabetes medication fills were assessed within 1 year before death, examining changes across three time periods: 12 to 8 months, 8 to 4 months, and 4 to 0 months before death.
TAKEAWAY:
The proportion of older patients receiving antidiabetes medications increased slightly from 71.4% in 2015 to 72.9% in 2019, with metformin showing the largest increase from 40.7% to 46.5% (standardized mean difference [SMD], −0.12) and sulfonylureas showing the largest decrease from 37.0% to 31.8% (SMD, 0.11).
The use of newer diabetes medications with cardiovascular benefits, such as GLP-1 receptor agonists and SGLT2 inhibitors, remained less common but showed increasing trends over time.
The use of any antidiabetes medication decreased from 66.1% in the 9 to 12 months before death to 60.8% in the last 4 months of life (P < .01), primarily due to the reduced use of metformin and sulfonylureas.
The use of both short-acting and long-acting insulin agents increased toward the end of life (from 28.0% to 32.9% and from 41.2% to 43.9%, respectively; both P < .001) , particularly among frailer individuals.
IN PRACTICE:
“[The study] findings underscore important implications for diabetes management in patients nearing the end of life. With ~70% of patients with T2D using at least one antidiabetes medication, there is a need to consider further de-escalation or deprescribing in this vulnerable population,” the authors wrote.
SOURCE:
The study was led by Alexander Kutz, MD, MPH, MSc, Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, and was published online in Diabetes Care.
LIMITATIONS:
The study lacked details on the reasons for changes in medication patterns, making it unclear whether these changes were due to clinical guidelines or to reduce adverse events. Moreover, the study could not capture transitions or substitutions between medications, information on the dosage data, and causes of death.
DISCLOSURES:
This study was supported by the Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School. Some authors reported receiving personal fees or research grants from the National Institutes of Health and other institutions and a few pharmaceutical companies. One author reported acting as a principal investigator and receiving a research grant from Boehringer-Ingelheim, unrelated to the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Retatrutide Produces Greatest Weight Loss
A systematic review of 26 randomized controlled trials (RCTs) finds that, among glucagon-like peptide 1 (GLP-1) receptor agonists and co-agonists on the market or still being investigated, the experimental drug retatrutide (Eli Lilly) produces the greatest weight loss.
The review, conducted by researchers at McGill University, Montreal, Quebec, Canada, examined three commercially available medications in the class and nine that have not yet received regulatory approval.
In healthy adults with overweight or obesity who did not have diabetes, the highest mean reductions in relative and absolute body weight were achieved with once-weekly triple glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 and glucagon receptor agonist retatrutide, followed by the dual GIP/GLP-1 agonist tirzepatide (Eli Lilly) and lastly by the GLP-1 agonist semaglutide (Novo Nordisk), according to the authors.
The use of all the GLP-1s or co-agonist medications “led to decreased body mass index (BMI), waist circumference, SBP (systolic blood pressure), and DBP (diastolic blood pressure),” wrote the authors in Annals of Internal Medicine. All the medications had a similar safety profile.
The researchers did not find any head-to-head studies, so instead examined the results from 26 RCTs that enrolled more than 15,000 patients. Only trials with a treatment duration of at least 16 weeks were included, to ensure that patients had at least a month of a fixed dose.
Not surprisingly, the review found that, except for semaglutide, trials with “dual and triple agonists generally reported numerically greater mean weight losses than single GLP-1 agonists.”
They caution, however, against drawing conclusions about comparative efficacy, as the populations, control groups, and contexts of the various studies might not be directly comparable. All the trial enrollees also received lifestyle modification along with drug therapy or placebo, but the interventions and protocols varied across the studies.
The authors found that individuals on retatrutide (12-mg once-weekly injection) lost 22% of body weight from baseline after 48 weeks. Tirzepatide (15 mg once-weekly injection) recipients lost almost 18% of body weight after 72 weeks, while those on semaglutide (2.4-mg once-weekly injection) lost about 14% after 68 weeks. Both tirzepatide and semaglutide are commercially available.
Patients taking liraglutide (3-mg once-daily injection), also on the market, lost up to 6% of body weight after 26 weeks.
The authors also examined studies of investigational agents and reported that the greatest loss, aside from retatrutide, was with the dual glucagon/GLP-1 agonists survodutide (Boehringer Ingelheim; 6%-15%) and mazdutide (Innovent Biologics; 7%-11%).
Orforglipron (Eli Lilly), a once-daily pill, produced weight loss of 9%-15%, depending on the dose.
The study found that four investigational drugs did not produce as much weight loss: Beinaglutide (0.2-mg injection three times daily, 6%), efpeglenatide (4- to 8-mg injection once weekly, about 7%), exenatide (10-mcg injection twice daily, 5-kg change in weight), and noiiglutide (once-daily injection, 9%).
The most common adverse events for all GLP-1s were gastrointestinal (GI), such as nausea, diarrhea, constipation, and vomiting. Across all agents, 60%-80% of patients taking the medications experienced a GI adverse event, although most were transient, according to the authors. A total of 6%-26% of patients discontinued treatment as a result of a side effect.
The authors said that no serious GI disorders, such as bowel obstruction or gastroparesis, were reported in any of the 26 trials.
The review also shows that it is likely that GLP-1s would have to be used chronically to have the greatest effect, said the authors. They noted that they found that trials “with longer treatment durations demonstrate similar weight loss results to those with shorter follow-up, reinforcing the idea that continuous treatment may be required.”
One coauthor reported receiving payments or honoraria from Boehringer Ingelheim, Eli Lilly, Janssen Pharmaceuticals, and Novo Nordisk. The study was carried out independently without any grant or other funding.
A version of this article first appeared on Medscape.com.
A systematic review of 26 randomized controlled trials (RCTs) finds that, among glucagon-like peptide 1 (GLP-1) receptor agonists and co-agonists on the market or still being investigated, the experimental drug retatrutide (Eli Lilly) produces the greatest weight loss.
The review, conducted by researchers at McGill University, Montreal, Quebec, Canada, examined three commercially available medications in the class and nine that have not yet received regulatory approval.
In healthy adults with overweight or obesity who did not have diabetes, the highest mean reductions in relative and absolute body weight were achieved with once-weekly triple glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 and glucagon receptor agonist retatrutide, followed by the dual GIP/GLP-1 agonist tirzepatide (Eli Lilly) and lastly by the GLP-1 agonist semaglutide (Novo Nordisk), according to the authors.
The use of all the GLP-1s or co-agonist medications “led to decreased body mass index (BMI), waist circumference, SBP (systolic blood pressure), and DBP (diastolic blood pressure),” wrote the authors in Annals of Internal Medicine. All the medications had a similar safety profile.
The researchers did not find any head-to-head studies, so instead examined the results from 26 RCTs that enrolled more than 15,000 patients. Only trials with a treatment duration of at least 16 weeks were included, to ensure that patients had at least a month of a fixed dose.
Not surprisingly, the review found that, except for semaglutide, trials with “dual and triple agonists generally reported numerically greater mean weight losses than single GLP-1 agonists.”
They caution, however, against drawing conclusions about comparative efficacy, as the populations, control groups, and contexts of the various studies might not be directly comparable. All the trial enrollees also received lifestyle modification along with drug therapy or placebo, but the interventions and protocols varied across the studies.
The authors found that individuals on retatrutide (12-mg once-weekly injection) lost 22% of body weight from baseline after 48 weeks. Tirzepatide (15 mg once-weekly injection) recipients lost almost 18% of body weight after 72 weeks, while those on semaglutide (2.4-mg once-weekly injection) lost about 14% after 68 weeks. Both tirzepatide and semaglutide are commercially available.
Patients taking liraglutide (3-mg once-daily injection), also on the market, lost up to 6% of body weight after 26 weeks.
The authors also examined studies of investigational agents and reported that the greatest loss, aside from retatrutide, was with the dual glucagon/GLP-1 agonists survodutide (Boehringer Ingelheim; 6%-15%) and mazdutide (Innovent Biologics; 7%-11%).
Orforglipron (Eli Lilly), a once-daily pill, produced weight loss of 9%-15%, depending on the dose.
The study found that four investigational drugs did not produce as much weight loss: Beinaglutide (0.2-mg injection three times daily, 6%), efpeglenatide (4- to 8-mg injection once weekly, about 7%), exenatide (10-mcg injection twice daily, 5-kg change in weight), and noiiglutide (once-daily injection, 9%).
The most common adverse events for all GLP-1s were gastrointestinal (GI), such as nausea, diarrhea, constipation, and vomiting. Across all agents, 60%-80% of patients taking the medications experienced a GI adverse event, although most were transient, according to the authors. A total of 6%-26% of patients discontinued treatment as a result of a side effect.
The authors said that no serious GI disorders, such as bowel obstruction or gastroparesis, were reported in any of the 26 trials.
The review also shows that it is likely that GLP-1s would have to be used chronically to have the greatest effect, said the authors. They noted that they found that trials “with longer treatment durations demonstrate similar weight loss results to those with shorter follow-up, reinforcing the idea that continuous treatment may be required.”
One coauthor reported receiving payments or honoraria from Boehringer Ingelheim, Eli Lilly, Janssen Pharmaceuticals, and Novo Nordisk. The study was carried out independently without any grant or other funding.
A version of this article first appeared on Medscape.com.
A systematic review of 26 randomized controlled trials (RCTs) finds that, among glucagon-like peptide 1 (GLP-1) receptor agonists and co-agonists on the market or still being investigated, the experimental drug retatrutide (Eli Lilly) produces the greatest weight loss.
The review, conducted by researchers at McGill University, Montreal, Quebec, Canada, examined three commercially available medications in the class and nine that have not yet received regulatory approval.
In healthy adults with overweight or obesity who did not have diabetes, the highest mean reductions in relative and absolute body weight were achieved with once-weekly triple glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 and glucagon receptor agonist retatrutide, followed by the dual GIP/GLP-1 agonist tirzepatide (Eli Lilly) and lastly by the GLP-1 agonist semaglutide (Novo Nordisk), according to the authors.
The use of all the GLP-1s or co-agonist medications “led to decreased body mass index (BMI), waist circumference, SBP (systolic blood pressure), and DBP (diastolic blood pressure),” wrote the authors in Annals of Internal Medicine. All the medications had a similar safety profile.
The researchers did not find any head-to-head studies, so instead examined the results from 26 RCTs that enrolled more than 15,000 patients. Only trials with a treatment duration of at least 16 weeks were included, to ensure that patients had at least a month of a fixed dose.
Not surprisingly, the review found that, except for semaglutide, trials with “dual and triple agonists generally reported numerically greater mean weight losses than single GLP-1 agonists.”
They caution, however, against drawing conclusions about comparative efficacy, as the populations, control groups, and contexts of the various studies might not be directly comparable. All the trial enrollees also received lifestyle modification along with drug therapy or placebo, but the interventions and protocols varied across the studies.
The authors found that individuals on retatrutide (12-mg once-weekly injection) lost 22% of body weight from baseline after 48 weeks. Tirzepatide (15 mg once-weekly injection) recipients lost almost 18% of body weight after 72 weeks, while those on semaglutide (2.4-mg once-weekly injection) lost about 14% after 68 weeks. Both tirzepatide and semaglutide are commercially available.
Patients taking liraglutide (3-mg once-daily injection), also on the market, lost up to 6% of body weight after 26 weeks.
The authors also examined studies of investigational agents and reported that the greatest loss, aside from retatrutide, was with the dual glucagon/GLP-1 agonists survodutide (Boehringer Ingelheim; 6%-15%) and mazdutide (Innovent Biologics; 7%-11%).
Orforglipron (Eli Lilly), a once-daily pill, produced weight loss of 9%-15%, depending on the dose.
The study found that four investigational drugs did not produce as much weight loss: Beinaglutide (0.2-mg injection three times daily, 6%), efpeglenatide (4- to 8-mg injection once weekly, about 7%), exenatide (10-mcg injection twice daily, 5-kg change in weight), and noiiglutide (once-daily injection, 9%).
The most common adverse events for all GLP-1s were gastrointestinal (GI), such as nausea, diarrhea, constipation, and vomiting. Across all agents, 60%-80% of patients taking the medications experienced a GI adverse event, although most were transient, according to the authors. A total of 6%-26% of patients discontinued treatment as a result of a side effect.
The authors said that no serious GI disorders, such as bowel obstruction or gastroparesis, were reported in any of the 26 trials.
The review also shows that it is likely that GLP-1s would have to be used chronically to have the greatest effect, said the authors. They noted that they found that trials “with longer treatment durations demonstrate similar weight loss results to those with shorter follow-up, reinforcing the idea that continuous treatment may be required.”
One coauthor reported receiving payments or honoraria from Boehringer Ingelheim, Eli Lilly, Janssen Pharmaceuticals, and Novo Nordisk. The study was carried out independently without any grant or other funding.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Using GLP-1s to Meet BMI Goal for Orthopedic Surgery
The woman, in severe pain from hip and knee osteoarthritis, was confined to a wheelchair and had been told that would likely be for life. To qualify for hip replacement surgery, she needed to lose 100 pounds, a seemingly impossible goal. But she wanted to try.
“We tried a couple of medicines — oral medicines off-label — topiramate, phentermine,” said Leslie Golden, MD, MPH, DABM, a family medicine physician and obesity medicine specialist in Watertown, Wisconsin, 42 miles northeast of Madison.
They weren’t enough. But then Golden turned to glucagon-like peptide 1 (GLP-1) receptor agonists, and they delivered.
“She did lose a significant amount of weight and was able to get the hip replacement,” said Golden.
It took a couple of years. However, seeing her walk into her office, rather than wheel in, “is still one of the joys of my practice,” Golden said. “She’s so grateful. She felt everyone else had written her off.”
As she told Golden: “If I fell and broke my leg today, they would take me to surgery without concern.”
Because her hip replacement was viewed as a nonemergency procedure, the accepted threshold for elective safe surgery was a body mass index (BMI) < 40. That BMI cutoff can vary from provider to provider and medical facility to medical facility but is often required for other surgeries as well, including kidney and lung transplants, gender-affirming surgery, bariatric surgery, hernia surgery, and in vitro fertilization procedures.
She worked with Rajit Chakravarty, MD, an adult reconstructive surgeon who practices in Watertown and nearby Madison, to oversee the weight loss.
High BMIs & Surgery Issues
High BMIs have long been linked with postsurgery complications, poor wound healing, and other issues, although some research now is questioning some of those associations. Even so, surgeons have long stressed weight loss for their patients with obesity before orthopedic and other procedures.
These days, surgeons are more likely to need to have that talk. In the last decade, the age-adjusted prevalence of severe obesity — a BMI of ≥ 40 — has increased from 7.7% to 9.7% of US adults. The number of joint replacements is also rising — more than 700,000 total knee arthroplasty (TKA) and more than 450,000 total hip arthroplasty (THA), according to the American Academy of Orthopaedic Surgeons. As the population ages, those numbers are expected to increase.
Making the GLP-1 Choice
GLP-1s aren’t the only choice, of course. But they’re often more effective, as Golden found, than other medications. And when his patients with obesity are offered bariatric surgery or GLP-1s, “people definitely want to avoid the bariatric surgery,” Chakravarty said.
With the Food and Drug Administration (FDA) approval of semaglutide (Wegovy) in June 2021 for chronic weight management and then tirzepatide (Zepbound) in November 2023, interest has boomed, he said, among his surgery candidates with a high BMI.
The FDA approved Wegovy based on clinical trials, including one in which participants lost an average of 12.4% of initial body weight compared with those on placebo. It approved Zepbound based on clinical trials, including one in which those on Zepbound lost an average of 18% of their body weight, compared with those on placebo.
The wheelchair-bound woman, now 65, began with a BMI of 63, Golden said. She negotiated a cutoff of 45 with the surgeon and got the go-ahead. Currently, her BMI is 36 as she stayed on the medications.
Beyond the benefit of GLP-1s helping patients meet the BMI cutoff, some research finds fewer postoperative infections and readmissions with their use. This study found the medications did lower both, and another found reduced readmissions and complications.
Growing Partnerships, Increasing Success
Helping patients lose weight isn’t just about lowering the BMI, Chakravarty pointed out. The aim is to improve nutritional health — to teach patients how to eat healthfully for their needs, in turn improving other health barometers. Referring them to an obesity medicine physician helps to meet those goals.
When Daniel Wiznia, MD, a Yale Medicine orthopedic surgeon and codirector of the Avascular Necrosis Program, has a patient who must delay a TKA or THA until they meet a BMI cutoff, he refers that patient to the Yale Medicine Center for Weight Management, New Haven, Connecticut, to learn about weight loss, including the options of anti-obesity medications or bariatric surgery.
Taking the GLP-1s can be a game changer, according to Wiznia and John Morton, MD, MPH, FACS, FASMBS, Yale’s medical director of Bariatric Surgery and professor and vice chair of surgery, who is a physician-director of the center. The program includes other options, such as bariatric surgery, and emphasizes diet and other lifestyle measures. GLP-1s give about a 15% weight loss, Morton said, compared with bariatric surgery providing up to 30%.
Sarah Stombaugh, MD, a family medicine and obesity medicine physician in Charlottesville, Virginia, often gets referrals from two orthopedic surgeons in her community. One recent patient in her early 60s had a BMI of 43.2, too high to qualify for the TKA she needed. On GLP-1s, the initial goal was to decrease a weight of 244 to 225, bringing the BMI to 39.9. The woman did that, then kept losing before her surgery was scheduled, getting to a weight of 210 or a BMI of 37 and staying there for 3 months before the surgery.
She had the TKA, and 5 months out, she is doing well, Stombaugh said. “We do medical weight loss primarily with the GLP-1s because they’re simply the best, the most effective,” Stombaugh said. She does occasionally use oral medications such as naltrexone/bupropion (Contrave).
Stombaugh sees the collaborating trend as still evolving. When she attends obesity medicine conferences, not all her colleagues report they are partnering with surgeons. But she predicts the practice will increase, saying the popularization of what she terms the more effective GLP-1 medications Wegovy and Zepbound is driving it. Partnering with the surgeon requires a conversation at the beginning, when the referral is made, about goals. After that, she sees her patient monthly and sends progress notes to the surgeon.
Golden collaborates with three orthopedic groups in her area, primarily for knee and hip surgeries, but has also helped patients meet the BMI cutoff before spine-related surgeries. She is helping a lung transplant patient now. She has seen several patients who must meet BMI requirements before starting in vitro fertilization, due to the need for conscious sedation for egg retrieval. She has had a few patients who had to meet a BMI cutoff for nonemergency hernia repair.
Insurance Issues
Insurance remains an issue for the pricey medications. “Only about a third of patients are routinely covered with insurance,” Morton said.
However, it’s improving, he said. Golden also finds about a third of private payers cover the medication but tries to use manufacturers’ coupons to help defray the costs (from about $1000 or $1400 to about $500 a month). She has sometimes gotten enough samples to get patients to their BMI goal
Morton consulted for Novo Nordisk, Eli Lilly, Olympus, Teleflex, and Johnson & Johnson.
A version of this article appeared on Medscape.com.
The woman, in severe pain from hip and knee osteoarthritis, was confined to a wheelchair and had been told that would likely be for life. To qualify for hip replacement surgery, she needed to lose 100 pounds, a seemingly impossible goal. But she wanted to try.
“We tried a couple of medicines — oral medicines off-label — topiramate, phentermine,” said Leslie Golden, MD, MPH, DABM, a family medicine physician and obesity medicine specialist in Watertown, Wisconsin, 42 miles northeast of Madison.
They weren’t enough. But then Golden turned to glucagon-like peptide 1 (GLP-1) receptor agonists, and they delivered.
“She did lose a significant amount of weight and was able to get the hip replacement,” said Golden.
It took a couple of years. However, seeing her walk into her office, rather than wheel in, “is still one of the joys of my practice,” Golden said. “She’s so grateful. She felt everyone else had written her off.”
As she told Golden: “If I fell and broke my leg today, they would take me to surgery without concern.”
Because her hip replacement was viewed as a nonemergency procedure, the accepted threshold for elective safe surgery was a body mass index (BMI) < 40. That BMI cutoff can vary from provider to provider and medical facility to medical facility but is often required for other surgeries as well, including kidney and lung transplants, gender-affirming surgery, bariatric surgery, hernia surgery, and in vitro fertilization procedures.
She worked with Rajit Chakravarty, MD, an adult reconstructive surgeon who practices in Watertown and nearby Madison, to oversee the weight loss.
High BMIs & Surgery Issues
High BMIs have long been linked with postsurgery complications, poor wound healing, and other issues, although some research now is questioning some of those associations. Even so, surgeons have long stressed weight loss for their patients with obesity before orthopedic and other procedures.
These days, surgeons are more likely to need to have that talk. In the last decade, the age-adjusted prevalence of severe obesity — a BMI of ≥ 40 — has increased from 7.7% to 9.7% of US adults. The number of joint replacements is also rising — more than 700,000 total knee arthroplasty (TKA) and more than 450,000 total hip arthroplasty (THA), according to the American Academy of Orthopaedic Surgeons. As the population ages, those numbers are expected to increase.
Making the GLP-1 Choice
GLP-1s aren’t the only choice, of course. But they’re often more effective, as Golden found, than other medications. And when his patients with obesity are offered bariatric surgery or GLP-1s, “people definitely want to avoid the bariatric surgery,” Chakravarty said.
With the Food and Drug Administration (FDA) approval of semaglutide (Wegovy) in June 2021 for chronic weight management and then tirzepatide (Zepbound) in November 2023, interest has boomed, he said, among his surgery candidates with a high BMI.
The FDA approved Wegovy based on clinical trials, including one in which participants lost an average of 12.4% of initial body weight compared with those on placebo. It approved Zepbound based on clinical trials, including one in which those on Zepbound lost an average of 18% of their body weight, compared with those on placebo.
The wheelchair-bound woman, now 65, began with a BMI of 63, Golden said. She negotiated a cutoff of 45 with the surgeon and got the go-ahead. Currently, her BMI is 36 as she stayed on the medications.
Beyond the benefit of GLP-1s helping patients meet the BMI cutoff, some research finds fewer postoperative infections and readmissions with their use. This study found the medications did lower both, and another found reduced readmissions and complications.
Growing Partnerships, Increasing Success
Helping patients lose weight isn’t just about lowering the BMI, Chakravarty pointed out. The aim is to improve nutritional health — to teach patients how to eat healthfully for their needs, in turn improving other health barometers. Referring them to an obesity medicine physician helps to meet those goals.
When Daniel Wiznia, MD, a Yale Medicine orthopedic surgeon and codirector of the Avascular Necrosis Program, has a patient who must delay a TKA or THA until they meet a BMI cutoff, he refers that patient to the Yale Medicine Center for Weight Management, New Haven, Connecticut, to learn about weight loss, including the options of anti-obesity medications or bariatric surgery.
Taking the GLP-1s can be a game changer, according to Wiznia and John Morton, MD, MPH, FACS, FASMBS, Yale’s medical director of Bariatric Surgery and professor and vice chair of surgery, who is a physician-director of the center. The program includes other options, such as bariatric surgery, and emphasizes diet and other lifestyle measures. GLP-1s give about a 15% weight loss, Morton said, compared with bariatric surgery providing up to 30%.
Sarah Stombaugh, MD, a family medicine and obesity medicine physician in Charlottesville, Virginia, often gets referrals from two orthopedic surgeons in her community. One recent patient in her early 60s had a BMI of 43.2, too high to qualify for the TKA she needed. On GLP-1s, the initial goal was to decrease a weight of 244 to 225, bringing the BMI to 39.9. The woman did that, then kept losing before her surgery was scheduled, getting to a weight of 210 or a BMI of 37 and staying there for 3 months before the surgery.
She had the TKA, and 5 months out, she is doing well, Stombaugh said. “We do medical weight loss primarily with the GLP-1s because they’re simply the best, the most effective,” Stombaugh said. She does occasionally use oral medications such as naltrexone/bupropion (Contrave).
Stombaugh sees the collaborating trend as still evolving. When she attends obesity medicine conferences, not all her colleagues report they are partnering with surgeons. But she predicts the practice will increase, saying the popularization of what she terms the more effective GLP-1 medications Wegovy and Zepbound is driving it. Partnering with the surgeon requires a conversation at the beginning, when the referral is made, about goals. After that, she sees her patient monthly and sends progress notes to the surgeon.
Golden collaborates with three orthopedic groups in her area, primarily for knee and hip surgeries, but has also helped patients meet the BMI cutoff before spine-related surgeries. She is helping a lung transplant patient now. She has seen several patients who must meet BMI requirements before starting in vitro fertilization, due to the need for conscious sedation for egg retrieval. She has had a few patients who had to meet a BMI cutoff for nonemergency hernia repair.
Insurance Issues
Insurance remains an issue for the pricey medications. “Only about a third of patients are routinely covered with insurance,” Morton said.
However, it’s improving, he said. Golden also finds about a third of private payers cover the medication but tries to use manufacturers’ coupons to help defray the costs (from about $1000 or $1400 to about $500 a month). She has sometimes gotten enough samples to get patients to their BMI goal
Morton consulted for Novo Nordisk, Eli Lilly, Olympus, Teleflex, and Johnson & Johnson.
A version of this article appeared on Medscape.com.
The woman, in severe pain from hip and knee osteoarthritis, was confined to a wheelchair and had been told that would likely be for life. To qualify for hip replacement surgery, she needed to lose 100 pounds, a seemingly impossible goal. But she wanted to try.
“We tried a couple of medicines — oral medicines off-label — topiramate, phentermine,” said Leslie Golden, MD, MPH, DABM, a family medicine physician and obesity medicine specialist in Watertown, Wisconsin, 42 miles northeast of Madison.
They weren’t enough. But then Golden turned to glucagon-like peptide 1 (GLP-1) receptor agonists, and they delivered.
“She did lose a significant amount of weight and was able to get the hip replacement,” said Golden.
It took a couple of years. However, seeing her walk into her office, rather than wheel in, “is still one of the joys of my practice,” Golden said. “She’s so grateful. She felt everyone else had written her off.”
As she told Golden: “If I fell and broke my leg today, they would take me to surgery without concern.”
Because her hip replacement was viewed as a nonemergency procedure, the accepted threshold for elective safe surgery was a body mass index (BMI) < 40. That BMI cutoff can vary from provider to provider and medical facility to medical facility but is often required for other surgeries as well, including kidney and lung transplants, gender-affirming surgery, bariatric surgery, hernia surgery, and in vitro fertilization procedures.
She worked with Rajit Chakravarty, MD, an adult reconstructive surgeon who practices in Watertown and nearby Madison, to oversee the weight loss.
High BMIs & Surgery Issues
High BMIs have long been linked with postsurgery complications, poor wound healing, and other issues, although some research now is questioning some of those associations. Even so, surgeons have long stressed weight loss for their patients with obesity before orthopedic and other procedures.
These days, surgeons are more likely to need to have that talk. In the last decade, the age-adjusted prevalence of severe obesity — a BMI of ≥ 40 — has increased from 7.7% to 9.7% of US adults. The number of joint replacements is also rising — more than 700,000 total knee arthroplasty (TKA) and more than 450,000 total hip arthroplasty (THA), according to the American Academy of Orthopaedic Surgeons. As the population ages, those numbers are expected to increase.
Making the GLP-1 Choice
GLP-1s aren’t the only choice, of course. But they’re often more effective, as Golden found, than other medications. And when his patients with obesity are offered bariatric surgery or GLP-1s, “people definitely want to avoid the bariatric surgery,” Chakravarty said.
With the Food and Drug Administration (FDA) approval of semaglutide (Wegovy) in June 2021 for chronic weight management and then tirzepatide (Zepbound) in November 2023, interest has boomed, he said, among his surgery candidates with a high BMI.
The FDA approved Wegovy based on clinical trials, including one in which participants lost an average of 12.4% of initial body weight compared with those on placebo. It approved Zepbound based on clinical trials, including one in which those on Zepbound lost an average of 18% of their body weight, compared with those on placebo.
The wheelchair-bound woman, now 65, began with a BMI of 63, Golden said. She negotiated a cutoff of 45 with the surgeon and got the go-ahead. Currently, her BMI is 36 as she stayed on the medications.
Beyond the benefit of GLP-1s helping patients meet the BMI cutoff, some research finds fewer postoperative infections and readmissions with their use. This study found the medications did lower both, and another found reduced readmissions and complications.
Growing Partnerships, Increasing Success
Helping patients lose weight isn’t just about lowering the BMI, Chakravarty pointed out. The aim is to improve nutritional health — to teach patients how to eat healthfully for their needs, in turn improving other health barometers. Referring them to an obesity medicine physician helps to meet those goals.
When Daniel Wiznia, MD, a Yale Medicine orthopedic surgeon and codirector of the Avascular Necrosis Program, has a patient who must delay a TKA or THA until they meet a BMI cutoff, he refers that patient to the Yale Medicine Center for Weight Management, New Haven, Connecticut, to learn about weight loss, including the options of anti-obesity medications or bariatric surgery.
Taking the GLP-1s can be a game changer, according to Wiznia and John Morton, MD, MPH, FACS, FASMBS, Yale’s medical director of Bariatric Surgery and professor and vice chair of surgery, who is a physician-director of the center. The program includes other options, such as bariatric surgery, and emphasizes diet and other lifestyle measures. GLP-1s give about a 15% weight loss, Morton said, compared with bariatric surgery providing up to 30%.
Sarah Stombaugh, MD, a family medicine and obesity medicine physician in Charlottesville, Virginia, often gets referrals from two orthopedic surgeons in her community. One recent patient in her early 60s had a BMI of 43.2, too high to qualify for the TKA she needed. On GLP-1s, the initial goal was to decrease a weight of 244 to 225, bringing the BMI to 39.9. The woman did that, then kept losing before her surgery was scheduled, getting to a weight of 210 or a BMI of 37 and staying there for 3 months before the surgery.
She had the TKA, and 5 months out, she is doing well, Stombaugh said. “We do medical weight loss primarily with the GLP-1s because they’re simply the best, the most effective,” Stombaugh said. She does occasionally use oral medications such as naltrexone/bupropion (Contrave).
Stombaugh sees the collaborating trend as still evolving. When she attends obesity medicine conferences, not all her colleagues report they are partnering with surgeons. But she predicts the practice will increase, saying the popularization of what she terms the more effective GLP-1 medications Wegovy and Zepbound is driving it. Partnering with the surgeon requires a conversation at the beginning, when the referral is made, about goals. After that, she sees her patient monthly and sends progress notes to the surgeon.
Golden collaborates with three orthopedic groups in her area, primarily for knee and hip surgeries, but has also helped patients meet the BMI cutoff before spine-related surgeries. She is helping a lung transplant patient now. She has seen several patients who must meet BMI requirements before starting in vitro fertilization, due to the need for conscious sedation for egg retrieval. She has had a few patients who had to meet a BMI cutoff for nonemergency hernia repair.
Insurance Issues
Insurance remains an issue for the pricey medications. “Only about a third of patients are routinely covered with insurance,” Morton said.
However, it’s improving, he said. Golden also finds about a third of private payers cover the medication but tries to use manufacturers’ coupons to help defray the costs (from about $1000 or $1400 to about $500 a month). She has sometimes gotten enough samples to get patients to their BMI goal
Morton consulted for Novo Nordisk, Eli Lilly, Olympus, Teleflex, and Johnson & Johnson.
A version of this article appeared on Medscape.com.
Exercising Longer May Boost Weight Loss, Meta-Analysis Shows
TOPLINE:
Aerobic exercise shows a linear relationship with weight loss, with 30 minutes of weekly exercise linked to reduced body weight, waist circumference, and body fat in adults who were overweight or had obesity.
METHODOLOGY:
- Researchers conducted a meta-analysis of randomized clinical trials to investigate the association of varying intensities and durations of aerobic exercise with adiposity measures in adults with obesity or who were overweight.
- Overall, 116 randomized clinical trials that spanned across North America, Asia, Europe, Australia, South America, and Africa and involved 6880 adults (mean age, 46 years; 61% women) were included.
- The trials were required to have intervention durations of at least 8 weeks; all trials used supervised aerobic exercise, such as walking or running, while the control groups remained sedentary or continued usual activities.
- The intensity of exercise was defined as: Light (40%-55% maximum heart rate), moderate (55%-70% maximum heart rate), and vigorous (70%-90% maximum heart rate).
- The primary outcomes were body weight changes and adverse events; the secondary outcomes included changes in waist circumference, quality-of-life scores, and reduction in medications like antihypertensives.
TAKEAWAY:
- Every 30 minutes of aerobic exercise per week was also associated with lower waist circumference (mean difference, −0.56 cm; 95% CI, –0.67 to –0.45), body fat percentage (mean difference, –0.37%; 95% CI, –0.43 to –0.31), and body fat mass (mean difference, –0.20 kg; 95% CI, –0.32 to –0.08), along with reduced visceral and subcutaneous adipose tissue.
- A dose-response meta-analysis revealed that body fat percentage improved most significantly with 150 minutes of aerobic exercise per week, while body weight and waist circumference decreased linearly with increasing duration of aerobic exercise at 300 min/wk at different intensities.
- Adverse events with aerobic exercise were mostly mild or moderate musculoskeletal symptoms.
IN PRACTICE:
“Point-specific estimates for different aerobic exercise duration and intensity can help patients and healthcare professionals select the optimal aerobic exercise duration and intensity according to their weight loss goals,” the authors wrote.
SOURCE:
The study was led by Ahmad Jayedi, PhD, of the Department of Epidemiology and Biostatistics in the School of Public Health at the Imperial College London in England. It was published online on December 26, 2024, in JAMA Network Open.
LIMITATIONS:
High heterogeneity was present in the data. Only one trial included measures of health-related quality of life, and two studies included measures of medication use. Dietary habits and smoking status of participants were not included in studies, so any potential effects were not risk adjusted for.
DISCLOSURES:
No funding sources were reported. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Aerobic exercise shows a linear relationship with weight loss, with 30 minutes of weekly exercise linked to reduced body weight, waist circumference, and body fat in adults who were overweight or had obesity.
METHODOLOGY:
- Researchers conducted a meta-analysis of randomized clinical trials to investigate the association of varying intensities and durations of aerobic exercise with adiposity measures in adults with obesity or who were overweight.
- Overall, 116 randomized clinical trials that spanned across North America, Asia, Europe, Australia, South America, and Africa and involved 6880 adults (mean age, 46 years; 61% women) were included.
- The trials were required to have intervention durations of at least 8 weeks; all trials used supervised aerobic exercise, such as walking or running, while the control groups remained sedentary or continued usual activities.
- The intensity of exercise was defined as: Light (40%-55% maximum heart rate), moderate (55%-70% maximum heart rate), and vigorous (70%-90% maximum heart rate).
- The primary outcomes were body weight changes and adverse events; the secondary outcomes included changes in waist circumference, quality-of-life scores, and reduction in medications like antihypertensives.
TAKEAWAY:
- Every 30 minutes of aerobic exercise per week was also associated with lower waist circumference (mean difference, −0.56 cm; 95% CI, –0.67 to –0.45), body fat percentage (mean difference, –0.37%; 95% CI, –0.43 to –0.31), and body fat mass (mean difference, –0.20 kg; 95% CI, –0.32 to –0.08), along with reduced visceral and subcutaneous adipose tissue.
- A dose-response meta-analysis revealed that body fat percentage improved most significantly with 150 minutes of aerobic exercise per week, while body weight and waist circumference decreased linearly with increasing duration of aerobic exercise at 300 min/wk at different intensities.
- Adverse events with aerobic exercise were mostly mild or moderate musculoskeletal symptoms.
IN PRACTICE:
“Point-specific estimates for different aerobic exercise duration and intensity can help patients and healthcare professionals select the optimal aerobic exercise duration and intensity according to their weight loss goals,” the authors wrote.
SOURCE:
The study was led by Ahmad Jayedi, PhD, of the Department of Epidemiology and Biostatistics in the School of Public Health at the Imperial College London in England. It was published online on December 26, 2024, in JAMA Network Open.
LIMITATIONS:
High heterogeneity was present in the data. Only one trial included measures of health-related quality of life, and two studies included measures of medication use. Dietary habits and smoking status of participants were not included in studies, so any potential effects were not risk adjusted for.
DISCLOSURES:
No funding sources were reported. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Aerobic exercise shows a linear relationship with weight loss, with 30 minutes of weekly exercise linked to reduced body weight, waist circumference, and body fat in adults who were overweight or had obesity.
METHODOLOGY:
- Researchers conducted a meta-analysis of randomized clinical trials to investigate the association of varying intensities and durations of aerobic exercise with adiposity measures in adults with obesity or who were overweight.
- Overall, 116 randomized clinical trials that spanned across North America, Asia, Europe, Australia, South America, and Africa and involved 6880 adults (mean age, 46 years; 61% women) were included.
- The trials were required to have intervention durations of at least 8 weeks; all trials used supervised aerobic exercise, such as walking or running, while the control groups remained sedentary or continued usual activities.
- The intensity of exercise was defined as: Light (40%-55% maximum heart rate), moderate (55%-70% maximum heart rate), and vigorous (70%-90% maximum heart rate).
- The primary outcomes were body weight changes and adverse events; the secondary outcomes included changes in waist circumference, quality-of-life scores, and reduction in medications like antihypertensives.
TAKEAWAY:
- Every 30 minutes of aerobic exercise per week was also associated with lower waist circumference (mean difference, −0.56 cm; 95% CI, –0.67 to –0.45), body fat percentage (mean difference, –0.37%; 95% CI, –0.43 to –0.31), and body fat mass (mean difference, –0.20 kg; 95% CI, –0.32 to –0.08), along with reduced visceral and subcutaneous adipose tissue.
- A dose-response meta-analysis revealed that body fat percentage improved most significantly with 150 minutes of aerobic exercise per week, while body weight and waist circumference decreased linearly with increasing duration of aerobic exercise at 300 min/wk at different intensities.
- Adverse events with aerobic exercise were mostly mild or moderate musculoskeletal symptoms.
IN PRACTICE:
“Point-specific estimates for different aerobic exercise duration and intensity can help patients and healthcare professionals select the optimal aerobic exercise duration and intensity according to their weight loss goals,” the authors wrote.
SOURCE:
The study was led by Ahmad Jayedi, PhD, of the Department of Epidemiology and Biostatistics in the School of Public Health at the Imperial College London in England. It was published online on December 26, 2024, in JAMA Network Open.
LIMITATIONS:
High heterogeneity was present in the data. Only one trial included measures of health-related quality of life, and two studies included measures of medication use. Dietary habits and smoking status of participants were not included in studies, so any potential effects were not risk adjusted for.
DISCLOSURES:
No funding sources were reported. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Exposure to Dioxins May Increase Obesity Risk
TOPLINE:
Combined exposure to dioxins and dioxin-like polychlorinated biphenyls (DL-PCBs) is significantly associated with an increased risk for obesity in adults, with 1,2,3,4,6,7,8-heptachlorodibenzo-p-dioxin (HpCDD) showing the greatest contribution.
METHODOLOGY:
- Researchers evaluated the relationship between mixed exposure to nine types of dioxins and DL-PCBs and obesity or obesity indices in 852 adults using data from the National Health and Nutrition Examination Survey from 2003 to 2004.
- They chose nine chemicals for analysis: 1,2,3,4,6,7,8-HpCDD; 1,2,3,4,6,7,8,9-octachlorodibenzo-p-dioxin (OCDD); 3,3’,4,4’,5-pentachlorodibenzofuran (PnCB); PCB28; PCB66; PCB74; PCB105; PCB118; and PCB156.
- General and abdominal obesity were present in 34% and 53.9% of participants, respectively.
- Multiple statistical approaches were employed to evaluate the association of exposures to dioxins and DL-PCBs with obesity. Mediation analysis was performed to assess the potential role of A1c in this association.
TAKEAWAY:
- Multivariable logistic regression analysis found that a single exposure to higher concentrations of 1,2,3,4,6,7,8-HpCDD; 1,2,3,4,6,7,8,9-OCDD; 3,3’,4,4’,5-PnCB; PCB74; PCB105; and PCB118 was associated with an increased risk for general and abdominal obesity (P for trend < .001 for all). A stratified analysis by sex found that except for PCB28, PCB66, PCB74, and PCB156, all chemicals were linked to increased general and abdominal obesity risk in both men and women.
- Combined exposure to dioxins and DL-PCBs was positively associated with the risk for obesity, with 1,2,3,4,6,7,8-HpCDD showing the greatest contribution.
- When considering obesity indices, 1,2,3,4,6,7,8,9-OCDD; 1,2,3,4,6,7,8-HpCDD; 3,3’,4,4’,5-PnCB; PCB74; PCB105; and PCB118 were significantly associated with body mass index and waist circumference.
- A1c levels significantly mediated the association between mixed exposure to dioxins and DL-PCBs and obesity (P < .05), with mediation proportions of 6.94% for general obesity and 5.21% for abdominal obesity.
IN PRACTICE:
“Our findings suggested that dioxins and DL-PCBs may be independent risk factors for obesity,” the authors wrote. “The hazards of dioxins on obesity should be emphasized, and additional studies are desirable to elucidate the potential mechanisms for dioxins on obesity in adults.”
SOURCE:
This study, led by Zhao-Xing Gao, Department of Epidemiology and Biostatistics, School of Public Health, Anhui Medical University and Center for Big Data and Population Health of IHM, both in Hefei, China, was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The cross-sectional nature of this study prevented the establishment of causal relationships between dioxins or DL-PCBs and obesity. This study relied on a small sample. Replacing chemical concentrations below the limit of detection with fixed values may have introduced bias.
DISCLOSURES:
This study was funded by grants from the National Natural Science Foundation of China, Research Fund of Anhui Institute of Translational Medicine, and Research Fund of Center for Big Data and Population Health of IHM. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Combined exposure to dioxins and dioxin-like polychlorinated biphenyls (DL-PCBs) is significantly associated with an increased risk for obesity in adults, with 1,2,3,4,6,7,8-heptachlorodibenzo-p-dioxin (HpCDD) showing the greatest contribution.
METHODOLOGY:
- Researchers evaluated the relationship between mixed exposure to nine types of dioxins and DL-PCBs and obesity or obesity indices in 852 adults using data from the National Health and Nutrition Examination Survey from 2003 to 2004.
- They chose nine chemicals for analysis: 1,2,3,4,6,7,8-HpCDD; 1,2,3,4,6,7,8,9-octachlorodibenzo-p-dioxin (OCDD); 3,3’,4,4’,5-pentachlorodibenzofuran (PnCB); PCB28; PCB66; PCB74; PCB105; PCB118; and PCB156.
- General and abdominal obesity were present in 34% and 53.9% of participants, respectively.
- Multiple statistical approaches were employed to evaluate the association of exposures to dioxins and DL-PCBs with obesity. Mediation analysis was performed to assess the potential role of A1c in this association.
TAKEAWAY:
- Multivariable logistic regression analysis found that a single exposure to higher concentrations of 1,2,3,4,6,7,8-HpCDD; 1,2,3,4,6,7,8,9-OCDD; 3,3’,4,4’,5-PnCB; PCB74; PCB105; and PCB118 was associated with an increased risk for general and abdominal obesity (P for trend < .001 for all). A stratified analysis by sex found that except for PCB28, PCB66, PCB74, and PCB156, all chemicals were linked to increased general and abdominal obesity risk in both men and women.
- Combined exposure to dioxins and DL-PCBs was positively associated with the risk for obesity, with 1,2,3,4,6,7,8-HpCDD showing the greatest contribution.
- When considering obesity indices, 1,2,3,4,6,7,8,9-OCDD; 1,2,3,4,6,7,8-HpCDD; 3,3’,4,4’,5-PnCB; PCB74; PCB105; and PCB118 were significantly associated with body mass index and waist circumference.
- A1c levels significantly mediated the association between mixed exposure to dioxins and DL-PCBs and obesity (P < .05), with mediation proportions of 6.94% for general obesity and 5.21% for abdominal obesity.
IN PRACTICE:
“Our findings suggested that dioxins and DL-PCBs may be independent risk factors for obesity,” the authors wrote. “The hazards of dioxins on obesity should be emphasized, and additional studies are desirable to elucidate the potential mechanisms for dioxins on obesity in adults.”
SOURCE:
This study, led by Zhao-Xing Gao, Department of Epidemiology and Biostatistics, School of Public Health, Anhui Medical University and Center for Big Data and Population Health of IHM, both in Hefei, China, was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The cross-sectional nature of this study prevented the establishment of causal relationships between dioxins or DL-PCBs and obesity. This study relied on a small sample. Replacing chemical concentrations below the limit of detection with fixed values may have introduced bias.
DISCLOSURES:
This study was funded by grants from the National Natural Science Foundation of China, Research Fund of Anhui Institute of Translational Medicine, and Research Fund of Center for Big Data and Population Health of IHM. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Combined exposure to dioxins and dioxin-like polychlorinated biphenyls (DL-PCBs) is significantly associated with an increased risk for obesity in adults, with 1,2,3,4,6,7,8-heptachlorodibenzo-p-dioxin (HpCDD) showing the greatest contribution.
METHODOLOGY:
- Researchers evaluated the relationship between mixed exposure to nine types of dioxins and DL-PCBs and obesity or obesity indices in 852 adults using data from the National Health and Nutrition Examination Survey from 2003 to 2004.
- They chose nine chemicals for analysis: 1,2,3,4,6,7,8-HpCDD; 1,2,3,4,6,7,8,9-octachlorodibenzo-p-dioxin (OCDD); 3,3’,4,4’,5-pentachlorodibenzofuran (PnCB); PCB28; PCB66; PCB74; PCB105; PCB118; and PCB156.
- General and abdominal obesity were present in 34% and 53.9% of participants, respectively.
- Multiple statistical approaches were employed to evaluate the association of exposures to dioxins and DL-PCBs with obesity. Mediation analysis was performed to assess the potential role of A1c in this association.
TAKEAWAY:
- Multivariable logistic regression analysis found that a single exposure to higher concentrations of 1,2,3,4,6,7,8-HpCDD; 1,2,3,4,6,7,8,9-OCDD; 3,3’,4,4’,5-PnCB; PCB74; PCB105; and PCB118 was associated with an increased risk for general and abdominal obesity (P for trend < .001 for all). A stratified analysis by sex found that except for PCB28, PCB66, PCB74, and PCB156, all chemicals were linked to increased general and abdominal obesity risk in both men and women.
- Combined exposure to dioxins and DL-PCBs was positively associated with the risk for obesity, with 1,2,3,4,6,7,8-HpCDD showing the greatest contribution.
- When considering obesity indices, 1,2,3,4,6,7,8,9-OCDD; 1,2,3,4,6,7,8-HpCDD; 3,3’,4,4’,5-PnCB; PCB74; PCB105; and PCB118 were significantly associated with body mass index and waist circumference.
- A1c levels significantly mediated the association between mixed exposure to dioxins and DL-PCBs and obesity (P < .05), with mediation proportions of 6.94% for general obesity and 5.21% for abdominal obesity.
IN PRACTICE:
“Our findings suggested that dioxins and DL-PCBs may be independent risk factors for obesity,” the authors wrote. “The hazards of dioxins on obesity should be emphasized, and additional studies are desirable to elucidate the potential mechanisms for dioxins on obesity in adults.”
SOURCE:
This study, led by Zhao-Xing Gao, Department of Epidemiology and Biostatistics, School of Public Health, Anhui Medical University and Center for Big Data and Population Health of IHM, both in Hefei, China, was published online in The Journal of Clinical Endocrinology & Metabolism.
LIMITATIONS:
The cross-sectional nature of this study prevented the establishment of causal relationships between dioxins or DL-PCBs and obesity. This study relied on a small sample. Replacing chemical concentrations below the limit of detection with fixed values may have introduced bias.
DISCLOSURES:
This study was funded by grants from the National Natural Science Foundation of China, Research Fund of Anhui Institute of Translational Medicine, and Research Fund of Center for Big Data and Population Health of IHM. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
GLP-1 RAs: When Not to Prescribe
December 31, 2024
This transcript has been edited for clarity.
I’m Tamaan K. Osbourne-Roberts, family medicine physician and lifestyle medicine physician, here to discuss GLP-1 receptor agonist (RA) contraindications — the skinny on when not to prescribe.
It can be hard not to think of GLP-1 RAs like Ozempic and Mounjaro as silver bullets, long-awaited miracle drugs that we should probably be putting in the water. And it’s true they have the potential to help a lot of people.
They include the following:
Patients with a family history of certain cancers. Given that GLP-1 RAs can increase the risk for thyroid cancer, patients with a personal or family history of medullary thyroid cancer or multiple endocrine neoplasia type 2 should not take these drugs.
Gut motility issues. Since one of the primary mechanisms of action for these drugs is to slow down the gut, patients with gastroparesis — diabetic or otherwise — or other gut motility issues should avoid these drugs. Patients with inflammatory bowel disease also should not use GLP-1 RAs.
Pancreatitis. These medications can increase the risk for serious pancreatitis on their own, so use in patients who have had pancreatitis already is not recommended.
Renal impairment. An eGFR [estimated glomerular filtrationrate] below threshold, typically around 30 mL/min per 1.73 m2, excludes GLP-1 RAs for some patients. Be certain to check the threshold for individual medications before prescribing.
And finally, pregnancy. These drugs generally should not be used in pregnancy, and people of childbearing age with the ability to become pregnant should use contraception while taking these medications.
GLP-1 RAs are great medications and have the potential to revolutionize obesity medicine, but like all drugs, it’s important to use them safely. Knowing when not to prescribe them is an important step in ensuring patient safety and will help ensure they are available for those who need them.
Tamaan K. Osbourne-Roberts, MD, MBA, Denver, Colorado, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
December 31, 2024
This transcript has been edited for clarity.
I’m Tamaan K. Osbourne-Roberts, family medicine physician and lifestyle medicine physician, here to discuss GLP-1 receptor agonist (RA) contraindications — the skinny on when not to prescribe.
It can be hard not to think of GLP-1 RAs like Ozempic and Mounjaro as silver bullets, long-awaited miracle drugs that we should probably be putting in the water. And it’s true they have the potential to help a lot of people.
They include the following:
Patients with a family history of certain cancers. Given that GLP-1 RAs can increase the risk for thyroid cancer, patients with a personal or family history of medullary thyroid cancer or multiple endocrine neoplasia type 2 should not take these drugs.
Gut motility issues. Since one of the primary mechanisms of action for these drugs is to slow down the gut, patients with gastroparesis — diabetic or otherwise — or other gut motility issues should avoid these drugs. Patients with inflammatory bowel disease also should not use GLP-1 RAs.
Pancreatitis. These medications can increase the risk for serious pancreatitis on their own, so use in patients who have had pancreatitis already is not recommended.
Renal impairment. An eGFR [estimated glomerular filtrationrate] below threshold, typically around 30 mL/min per 1.73 m2, excludes GLP-1 RAs for some patients. Be certain to check the threshold for individual medications before prescribing.
And finally, pregnancy. These drugs generally should not be used in pregnancy, and people of childbearing age with the ability to become pregnant should use contraception while taking these medications.
GLP-1 RAs are great medications and have the potential to revolutionize obesity medicine, but like all drugs, it’s important to use them safely. Knowing when not to prescribe them is an important step in ensuring patient safety and will help ensure they are available for those who need them.
Tamaan K. Osbourne-Roberts, MD, MBA, Denver, Colorado, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
December 31, 2024
This transcript has been edited for clarity.
I’m Tamaan K. Osbourne-Roberts, family medicine physician and lifestyle medicine physician, here to discuss GLP-1 receptor agonist (RA) contraindications — the skinny on when not to prescribe.
It can be hard not to think of GLP-1 RAs like Ozempic and Mounjaro as silver bullets, long-awaited miracle drugs that we should probably be putting in the water. And it’s true they have the potential to help a lot of people.
They include the following:
Patients with a family history of certain cancers. Given that GLP-1 RAs can increase the risk for thyroid cancer, patients with a personal or family history of medullary thyroid cancer or multiple endocrine neoplasia type 2 should not take these drugs.
Gut motility issues. Since one of the primary mechanisms of action for these drugs is to slow down the gut, patients with gastroparesis — diabetic or otherwise — or other gut motility issues should avoid these drugs. Patients with inflammatory bowel disease also should not use GLP-1 RAs.
Pancreatitis. These medications can increase the risk for serious pancreatitis on their own, so use in patients who have had pancreatitis already is not recommended.
Renal impairment. An eGFR [estimated glomerular filtrationrate] below threshold, typically around 30 mL/min per 1.73 m2, excludes GLP-1 RAs for some patients. Be certain to check the threshold for individual medications before prescribing.
And finally, pregnancy. These drugs generally should not be used in pregnancy, and people of childbearing age with the ability to become pregnant should use contraception while taking these medications.
GLP-1 RAs are great medications and have the potential to revolutionize obesity medicine, but like all drugs, it’s important to use them safely. Knowing when not to prescribe them is an important step in ensuring patient safety and will help ensure they are available for those who need them.
Tamaan K. Osbourne-Roberts, MD, MBA, Denver, Colorado, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Obesity Drug Zepbound Approved for Obstructive Sleep Apnea
The Food and Drug Administration (FDA) has approved the obesity treatment tirzepatide (Zepbound, Eli Lilly) for treating moderate to severe obstructive sleep apnea (OSA) in adults with obesity.
The new indication is for use in combination with reduced-calorie diet and increased physical activity. The once-weekly injectable is the first-ever drug treatment for OSA.
“Today’s approval marks the first drug treatment option for certain patients with obstructive sleep apnea,” said Sally Seymour, MD, director of the Division of Pulmonology, Allergy, and Critical Care in the FDA’s Center for Drug Evaluation and Research. “This is a major step forward for patients with obstructive sleep apnea.”
Excess weight is a major risk factor for OSA, in which the upper airways become blocked multiple times during sleep and obstruct breathing. The condition causes loud snoring, recurrent awakenings, and daytime sleepiness. It is also associated with cardiovascular disease.
Tirzepatide, a dual glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic polypeptide receptor agonist, was initially approved with brand name Mounjaro in May 2022 for the treatment of type 2 diabetes, and as Zepbound for weight loss in November 2023.
The new OSA approval was based on two phase 3, double-blind randomized controlled trials, SURMOUNT-OSA, in patients with obesity and moderate to severe OSA, conducted at 60 sites in nine countries. Results from both were presented in June 2024 at the annual Scientific Sessions of the American Diabetes Association and were simultaneously published in The New England Journal of Medicine.
The first trial enrolled 469 participants who were unable or unwilling to use PAP therapy, while the second included 234 who had been using PAP for at least 3 months and planned to continue during the trial. In both, the participants randomly received either 10 mg or 15 mg of tirzepatide or placebo once weekly for 52 weeks.
At baseline, 65%-70% of participants had severe OSA, with more than 30 events/h on the apnea-hypopnea index (AHI) and a mean of 51.5 events/h. By 52 weeks, those randomized to tirzepatide had 27-30 fewer events/h, compared with 4-6 fewer events/h for those taking placebo. In addition, significantly more of those on tirzepatide achieved OSA remission or severity reduction to mild.
Those randomized to tirzepatide also averaged up to 20% weight loss, significantly more than with placebo. “The improvement in AHI in participants with OSA is likely related to body weight reduction with Zepbound,” according to an FDA statement.
Side effects of tirzepatide include nausea, diarrhea, vomiting, constipation, abdominal discomfort and pain, injection site reactions, fatigue, hypersensitivity reactions (typically fever and rash), burping, hair loss, and gastroesophageal reflux disease.
In an editorial accompanying The New England Journal of Medicine publication of the SURMOUNT-OSA results, Sanjay R. Patel, MD, wrote: “The potential incorporation of tirzepatide into treatment algorithms for obstructive sleep apnea should include consideration of the challenges of adherence to treatment and the imperative to address racial disparities in medical care.”
Patel, who is professor of medicine and epidemiology at the University of Pittsburgh in Pennsylvania, and medical director of the University of Pittsburgh Medical Center’s Comprehensive Sleep Disorders program, pointed out that suboptimal adherence to continuous PAP therapy has been a major limitation, but that adherence to the GLP-1 drug class has also been suboptimal.
“Although adherence to tirzepatide therapy in the SURMOUNT-OSA trial was high, real-world evidence suggests that nearly 50% of patients who begin treatment with a GLP-1 receptor agonist for obesity discontinue therapy within 12 months. Thus, it is likely that any incorporation of tirzepatide into treatment pathways for obstructive sleep apnea will not diminish the importance of long-term strategies to optimize adherence to treatment.”
Moreover, Patel noted, “racial disparities in the use of GLP-1 receptor agonists among patients with diabetes arouse concern that the addition of tirzepatide as a treatment option for obstructive sleep apnea without directly addressing policies relative to coverage of care will only further exacerbate already pervasive disparities in clinical care for obstructive sleep apnea.”
Patel reported consulting for Apnimed, Bayer Pharmaceuticals, Lilly USA, NovaResp Technologies, Philips Respironics, and Powell Mansfield. He is a fiduciary officer of BreathPA.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (FDA) has approved the obesity treatment tirzepatide (Zepbound, Eli Lilly) for treating moderate to severe obstructive sleep apnea (OSA) in adults with obesity.
The new indication is for use in combination with reduced-calorie diet and increased physical activity. The once-weekly injectable is the first-ever drug treatment for OSA.
“Today’s approval marks the first drug treatment option for certain patients with obstructive sleep apnea,” said Sally Seymour, MD, director of the Division of Pulmonology, Allergy, and Critical Care in the FDA’s Center for Drug Evaluation and Research. “This is a major step forward for patients with obstructive sleep apnea.”
Excess weight is a major risk factor for OSA, in which the upper airways become blocked multiple times during sleep and obstruct breathing. The condition causes loud snoring, recurrent awakenings, and daytime sleepiness. It is also associated with cardiovascular disease.
Tirzepatide, a dual glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic polypeptide receptor agonist, was initially approved with brand name Mounjaro in May 2022 for the treatment of type 2 diabetes, and as Zepbound for weight loss in November 2023.
The new OSA approval was based on two phase 3, double-blind randomized controlled trials, SURMOUNT-OSA, in patients with obesity and moderate to severe OSA, conducted at 60 sites in nine countries. Results from both were presented in June 2024 at the annual Scientific Sessions of the American Diabetes Association and were simultaneously published in The New England Journal of Medicine.
The first trial enrolled 469 participants who were unable or unwilling to use PAP therapy, while the second included 234 who had been using PAP for at least 3 months and planned to continue during the trial. In both, the participants randomly received either 10 mg or 15 mg of tirzepatide or placebo once weekly for 52 weeks.
At baseline, 65%-70% of participants had severe OSA, with more than 30 events/h on the apnea-hypopnea index (AHI) and a mean of 51.5 events/h. By 52 weeks, those randomized to tirzepatide had 27-30 fewer events/h, compared with 4-6 fewer events/h for those taking placebo. In addition, significantly more of those on tirzepatide achieved OSA remission or severity reduction to mild.
Those randomized to tirzepatide also averaged up to 20% weight loss, significantly more than with placebo. “The improvement in AHI in participants with OSA is likely related to body weight reduction with Zepbound,” according to an FDA statement.
Side effects of tirzepatide include nausea, diarrhea, vomiting, constipation, abdominal discomfort and pain, injection site reactions, fatigue, hypersensitivity reactions (typically fever and rash), burping, hair loss, and gastroesophageal reflux disease.
In an editorial accompanying The New England Journal of Medicine publication of the SURMOUNT-OSA results, Sanjay R. Patel, MD, wrote: “The potential incorporation of tirzepatide into treatment algorithms for obstructive sleep apnea should include consideration of the challenges of adherence to treatment and the imperative to address racial disparities in medical care.”
Patel, who is professor of medicine and epidemiology at the University of Pittsburgh in Pennsylvania, and medical director of the University of Pittsburgh Medical Center’s Comprehensive Sleep Disorders program, pointed out that suboptimal adherence to continuous PAP therapy has been a major limitation, but that adherence to the GLP-1 drug class has also been suboptimal.
“Although adherence to tirzepatide therapy in the SURMOUNT-OSA trial was high, real-world evidence suggests that nearly 50% of patients who begin treatment with a GLP-1 receptor agonist for obesity discontinue therapy within 12 months. Thus, it is likely that any incorporation of tirzepatide into treatment pathways for obstructive sleep apnea will not diminish the importance of long-term strategies to optimize adherence to treatment.”
Moreover, Patel noted, “racial disparities in the use of GLP-1 receptor agonists among patients with diabetes arouse concern that the addition of tirzepatide as a treatment option for obstructive sleep apnea without directly addressing policies relative to coverage of care will only further exacerbate already pervasive disparities in clinical care for obstructive sleep apnea.”
Patel reported consulting for Apnimed, Bayer Pharmaceuticals, Lilly USA, NovaResp Technologies, Philips Respironics, and Powell Mansfield. He is a fiduciary officer of BreathPA.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (FDA) has approved the obesity treatment tirzepatide (Zepbound, Eli Lilly) for treating moderate to severe obstructive sleep apnea (OSA) in adults with obesity.
The new indication is for use in combination with reduced-calorie diet and increased physical activity. The once-weekly injectable is the first-ever drug treatment for OSA.
“Today’s approval marks the first drug treatment option for certain patients with obstructive sleep apnea,” said Sally Seymour, MD, director of the Division of Pulmonology, Allergy, and Critical Care in the FDA’s Center for Drug Evaluation and Research. “This is a major step forward for patients with obstructive sleep apnea.”
Excess weight is a major risk factor for OSA, in which the upper airways become blocked multiple times during sleep and obstruct breathing. The condition causes loud snoring, recurrent awakenings, and daytime sleepiness. It is also associated with cardiovascular disease.
Tirzepatide, a dual glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic polypeptide receptor agonist, was initially approved with brand name Mounjaro in May 2022 for the treatment of type 2 diabetes, and as Zepbound for weight loss in November 2023.
The new OSA approval was based on two phase 3, double-blind randomized controlled trials, SURMOUNT-OSA, in patients with obesity and moderate to severe OSA, conducted at 60 sites in nine countries. Results from both were presented in June 2024 at the annual Scientific Sessions of the American Diabetes Association and were simultaneously published in The New England Journal of Medicine.
The first trial enrolled 469 participants who were unable or unwilling to use PAP therapy, while the second included 234 who had been using PAP for at least 3 months and planned to continue during the trial. In both, the participants randomly received either 10 mg or 15 mg of tirzepatide or placebo once weekly for 52 weeks.
At baseline, 65%-70% of participants had severe OSA, with more than 30 events/h on the apnea-hypopnea index (AHI) and a mean of 51.5 events/h. By 52 weeks, those randomized to tirzepatide had 27-30 fewer events/h, compared with 4-6 fewer events/h for those taking placebo. In addition, significantly more of those on tirzepatide achieved OSA remission or severity reduction to mild.
Those randomized to tirzepatide also averaged up to 20% weight loss, significantly more than with placebo. “The improvement in AHI in participants with OSA is likely related to body weight reduction with Zepbound,” according to an FDA statement.
Side effects of tirzepatide include nausea, diarrhea, vomiting, constipation, abdominal discomfort and pain, injection site reactions, fatigue, hypersensitivity reactions (typically fever and rash), burping, hair loss, and gastroesophageal reflux disease.
In an editorial accompanying The New England Journal of Medicine publication of the SURMOUNT-OSA results, Sanjay R. Patel, MD, wrote: “The potential incorporation of tirzepatide into treatment algorithms for obstructive sleep apnea should include consideration of the challenges of adherence to treatment and the imperative to address racial disparities in medical care.”
Patel, who is professor of medicine and epidemiology at the University of Pittsburgh in Pennsylvania, and medical director of the University of Pittsburgh Medical Center’s Comprehensive Sleep Disorders program, pointed out that suboptimal adherence to continuous PAP therapy has been a major limitation, but that adherence to the GLP-1 drug class has also been suboptimal.
“Although adherence to tirzepatide therapy in the SURMOUNT-OSA trial was high, real-world evidence suggests that nearly 50% of patients who begin treatment with a GLP-1 receptor agonist for obesity discontinue therapy within 12 months. Thus, it is likely that any incorporation of tirzepatide into treatment pathways for obstructive sleep apnea will not diminish the importance of long-term strategies to optimize adherence to treatment.”
Moreover, Patel noted, “racial disparities in the use of GLP-1 receptor agonists among patients with diabetes arouse concern that the addition of tirzepatide as a treatment option for obstructive sleep apnea without directly addressing policies relative to coverage of care will only further exacerbate already pervasive disparities in clinical care for obstructive sleep apnea.”
Patel reported consulting for Apnimed, Bayer Pharmaceuticals, Lilly USA, NovaResp Technologies, Philips Respironics, and Powell Mansfield. He is a fiduciary officer of BreathPA.
A version of this article first appeared on Medscape.com.