User login
Two New Studies on Benzoyl Peroxide Provide Reassuring Data on Safety
Two .
Earlier this year, controversy erupted after an independent lab Valisure petitioned the US Food and Drug Administration (FDA) to recall acne products with BP because it found extremely high levels of the carcinogen benzene. In the research, the lab directors contended that the products can form over 800 times the “conditionally restricted” FDA concentration limit of 2 parts per million (ppm) of benzene, with both prescription and over-the-counter (OTC) products affected. The issue, according to the lab’s report, is one of degradation, not contamination; BP can decompose into benzene. Exposures to benzene have been linked with a higher risk for leukemia and other blood cancers.
(“Conditionally restricted” means that the maximum of 2 ppm only applies to a drug product in which the use of benzene is unavoidable in order to produce a drug product with a significant therapeutic advance, according to FDA guidance.)
Critics of the report questioned the method used to test the products, calling for more “real-world” use data, and said the temperature used may not be what is expected with everyday use.
Now, both new studies are reassuring about the safety of the products, John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said in a telephone interview. He was a coauthor of both studies. A leading dermatologist not involved in the new research reviewed the findings and agreed.
One study using data from the National Health and Nutrition Examination Survey compared blood levels of benzene between 14 people who had used BP products and 65 people without a history of BP product use, finding no difference between the groups .
The other, much larger study analyzed electronic health records of more than 27,000 patients with acne using BP products, comparing them with more than 27,000 controls who did not use the products. The patients were followed for 10 years after the use of BP products began, and no increased risk for cancer, either blood cancers or solid tumors, was found.
The studies were recently published in the Journal of the American Academy of Dermatology.
“Both studies are well done,” said Henry W. Lim, MD, former chair of the Department of Dermatology and senior vice president for academic affairs at Henry Ford Health, Detroit. Dr. Lim, a former president of the American Academy of Dermatology, reviewed the results of both studies.
“These studies indicate that [a] report of detection of benzene in [BP] products exposed to high temperature does not have any relevant clinical significance, both in terms of blood levels and in terms of internal cancer,” Dr. Lim said. “This is consistent with the clinical experience of practicing dermatologists; no internal side effects have been observed in patients using [BP products].”
Further Details
Under high temperatures, or over a long period, BP can decompose to benzene, a colorless, flammable liquid with a sweet odor. Benzene is formed from natural processes such as forest fires and volcanoes, according to the American Cancer Society, and is found in the air, cigarette smoke, some foods (at low levels), and contaminated drinking water. It’s one of the 20 widely used chemicals involved in making plastics, resins, detergents, and pesticides, among other products.
In the study evaluating blood levels, the researchers matched 14 people who used BP products currently with 65 controls who did not. Five (36%) of those using the products had detectable blood levels; 21 (32%) of those who did not use them did. There was no association between BP exposure and detectable blood benzene levels (odds ratio, 1.12; P = .80).
In the larger study, the researchers used the TriNetX US Collaborative Network database, comparing more than 27,000 patients treated with BP products for acne with more than 27,000 patients aged 12-40 years who had a diagnosis of nevus or seborrheic keratosis with no exposure to prescribed BP or any diagnosis of acne, hidradenitis suppurativa, or rosacea. The researchers looked at the database over the subsequent 10 years to determine the risk for either blood cancers or internal malignancies.
Compared with patients diagnosed with nevus or seborrheic keratosis, those with acne treated with BP had no significant difference in the risk for lymphoma (hazard ratio [HR], 1.00), leukemia (HR, 0.91), any lymphoma or leukemia (HR, 1.04), and internal malignancies (HR, 0.93).
The findings suggest no increased risk for malignancy, the researchers said, although they acknowledged study limitations, such as possible misclassification of BP exposure due to OTC availability and other issues.
Value of BP Treatments
BP is the “go-to” acne treatment, as Dr. Barbieri pointed out. “It’s probably the number one treatment for acne,” and there’s no substitute for it and it’s one of the most effective topical acne treatments, he noted.
Despite the reassuring findings, Dr. Barbieri repeated advice he gave soon after the Valisure report was released. Use common sense and don’t store BP-containing products in hot cars or other hot environments. In warmer climates, refrigeration could be considered, he said. Discard old products. Manufacturers should use cold-chain storage from the manufacturing site to retail or pharmacy sale sites, he added.
FDA and Citizen Petition Status
Asked about the status of the petition from Valisure, an FDA spokesperson said: “The FDA does not comment on the status of pending petitions.”
Dr. Barbieri and Dr. Lim had no relevant disclosures. There were no funding sources for either of the two studies.
A version of this article first appeared on Medscape.com.
Two .
Earlier this year, controversy erupted after an independent lab Valisure petitioned the US Food and Drug Administration (FDA) to recall acne products with BP because it found extremely high levels of the carcinogen benzene. In the research, the lab directors contended that the products can form over 800 times the “conditionally restricted” FDA concentration limit of 2 parts per million (ppm) of benzene, with both prescription and over-the-counter (OTC) products affected. The issue, according to the lab’s report, is one of degradation, not contamination; BP can decompose into benzene. Exposures to benzene have been linked with a higher risk for leukemia and other blood cancers.
(“Conditionally restricted” means that the maximum of 2 ppm only applies to a drug product in which the use of benzene is unavoidable in order to produce a drug product with a significant therapeutic advance, according to FDA guidance.)
Critics of the report questioned the method used to test the products, calling for more “real-world” use data, and said the temperature used may not be what is expected with everyday use.
Now, both new studies are reassuring about the safety of the products, John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said in a telephone interview. He was a coauthor of both studies. A leading dermatologist not involved in the new research reviewed the findings and agreed.
One study using data from the National Health and Nutrition Examination Survey compared blood levels of benzene between 14 people who had used BP products and 65 people without a history of BP product use, finding no difference between the groups .
The other, much larger study analyzed electronic health records of more than 27,000 patients with acne using BP products, comparing them with more than 27,000 controls who did not use the products. The patients were followed for 10 years after the use of BP products began, and no increased risk for cancer, either blood cancers or solid tumors, was found.
The studies were recently published in the Journal of the American Academy of Dermatology.
“Both studies are well done,” said Henry W. Lim, MD, former chair of the Department of Dermatology and senior vice president for academic affairs at Henry Ford Health, Detroit. Dr. Lim, a former president of the American Academy of Dermatology, reviewed the results of both studies.
“These studies indicate that [a] report of detection of benzene in [BP] products exposed to high temperature does not have any relevant clinical significance, both in terms of blood levels and in terms of internal cancer,” Dr. Lim said. “This is consistent with the clinical experience of practicing dermatologists; no internal side effects have been observed in patients using [BP products].”
Further Details
Under high temperatures, or over a long period, BP can decompose to benzene, a colorless, flammable liquid with a sweet odor. Benzene is formed from natural processes such as forest fires and volcanoes, according to the American Cancer Society, and is found in the air, cigarette smoke, some foods (at low levels), and contaminated drinking water. It’s one of the 20 widely used chemicals involved in making plastics, resins, detergents, and pesticides, among other products.
In the study evaluating blood levels, the researchers matched 14 people who used BP products currently with 65 controls who did not. Five (36%) of those using the products had detectable blood levels; 21 (32%) of those who did not use them did. There was no association between BP exposure and detectable blood benzene levels (odds ratio, 1.12; P = .80).
In the larger study, the researchers used the TriNetX US Collaborative Network database, comparing more than 27,000 patients treated with BP products for acne with more than 27,000 patients aged 12-40 years who had a diagnosis of nevus or seborrheic keratosis with no exposure to prescribed BP or any diagnosis of acne, hidradenitis suppurativa, or rosacea. The researchers looked at the database over the subsequent 10 years to determine the risk for either blood cancers or internal malignancies.
Compared with patients diagnosed with nevus or seborrheic keratosis, those with acne treated with BP had no significant difference in the risk for lymphoma (hazard ratio [HR], 1.00), leukemia (HR, 0.91), any lymphoma or leukemia (HR, 1.04), and internal malignancies (HR, 0.93).
The findings suggest no increased risk for malignancy, the researchers said, although they acknowledged study limitations, such as possible misclassification of BP exposure due to OTC availability and other issues.
Value of BP Treatments
BP is the “go-to” acne treatment, as Dr. Barbieri pointed out. “It’s probably the number one treatment for acne,” and there’s no substitute for it and it’s one of the most effective topical acne treatments, he noted.
Despite the reassuring findings, Dr. Barbieri repeated advice he gave soon after the Valisure report was released. Use common sense and don’t store BP-containing products in hot cars or other hot environments. In warmer climates, refrigeration could be considered, he said. Discard old products. Manufacturers should use cold-chain storage from the manufacturing site to retail or pharmacy sale sites, he added.
FDA and Citizen Petition Status
Asked about the status of the petition from Valisure, an FDA spokesperson said: “The FDA does not comment on the status of pending petitions.”
Dr. Barbieri and Dr. Lim had no relevant disclosures. There were no funding sources for either of the two studies.
A version of this article first appeared on Medscape.com.
Two .
Earlier this year, controversy erupted after an independent lab Valisure petitioned the US Food and Drug Administration (FDA) to recall acne products with BP because it found extremely high levels of the carcinogen benzene. In the research, the lab directors contended that the products can form over 800 times the “conditionally restricted” FDA concentration limit of 2 parts per million (ppm) of benzene, with both prescription and over-the-counter (OTC) products affected. The issue, according to the lab’s report, is one of degradation, not contamination; BP can decompose into benzene. Exposures to benzene have been linked with a higher risk for leukemia and other blood cancers.
(“Conditionally restricted” means that the maximum of 2 ppm only applies to a drug product in which the use of benzene is unavoidable in order to produce a drug product with a significant therapeutic advance, according to FDA guidance.)
Critics of the report questioned the method used to test the products, calling for more “real-world” use data, and said the temperature used may not be what is expected with everyday use.
Now, both new studies are reassuring about the safety of the products, John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said in a telephone interview. He was a coauthor of both studies. A leading dermatologist not involved in the new research reviewed the findings and agreed.
One study using data from the National Health and Nutrition Examination Survey compared blood levels of benzene between 14 people who had used BP products and 65 people without a history of BP product use, finding no difference between the groups .
The other, much larger study analyzed electronic health records of more than 27,000 patients with acne using BP products, comparing them with more than 27,000 controls who did not use the products. The patients were followed for 10 years after the use of BP products began, and no increased risk for cancer, either blood cancers or solid tumors, was found.
The studies were recently published in the Journal of the American Academy of Dermatology.
“Both studies are well done,” said Henry W. Lim, MD, former chair of the Department of Dermatology and senior vice president for academic affairs at Henry Ford Health, Detroit. Dr. Lim, a former president of the American Academy of Dermatology, reviewed the results of both studies.
“These studies indicate that [a] report of detection of benzene in [BP] products exposed to high temperature does not have any relevant clinical significance, both in terms of blood levels and in terms of internal cancer,” Dr. Lim said. “This is consistent with the clinical experience of practicing dermatologists; no internal side effects have been observed in patients using [BP products].”
Further Details
Under high temperatures, or over a long period, BP can decompose to benzene, a colorless, flammable liquid with a sweet odor. Benzene is formed from natural processes such as forest fires and volcanoes, according to the American Cancer Society, and is found in the air, cigarette smoke, some foods (at low levels), and contaminated drinking water. It’s one of the 20 widely used chemicals involved in making plastics, resins, detergents, and pesticides, among other products.
In the study evaluating blood levels, the researchers matched 14 people who used BP products currently with 65 controls who did not. Five (36%) of those using the products had detectable blood levels; 21 (32%) of those who did not use them did. There was no association between BP exposure and detectable blood benzene levels (odds ratio, 1.12; P = .80).
In the larger study, the researchers used the TriNetX US Collaborative Network database, comparing more than 27,000 patients treated with BP products for acne with more than 27,000 patients aged 12-40 years who had a diagnosis of nevus or seborrheic keratosis with no exposure to prescribed BP or any diagnosis of acne, hidradenitis suppurativa, or rosacea. The researchers looked at the database over the subsequent 10 years to determine the risk for either blood cancers or internal malignancies.
Compared with patients diagnosed with nevus or seborrheic keratosis, those with acne treated with BP had no significant difference in the risk for lymphoma (hazard ratio [HR], 1.00), leukemia (HR, 0.91), any lymphoma or leukemia (HR, 1.04), and internal malignancies (HR, 0.93).
The findings suggest no increased risk for malignancy, the researchers said, although they acknowledged study limitations, such as possible misclassification of BP exposure due to OTC availability and other issues.
Value of BP Treatments
BP is the “go-to” acne treatment, as Dr. Barbieri pointed out. “It’s probably the number one treatment for acne,” and there’s no substitute for it and it’s one of the most effective topical acne treatments, he noted.
Despite the reassuring findings, Dr. Barbieri repeated advice he gave soon after the Valisure report was released. Use common sense and don’t store BP-containing products in hot cars or other hot environments. In warmer climates, refrigeration could be considered, he said. Discard old products. Manufacturers should use cold-chain storage from the manufacturing site to retail or pharmacy sale sites, he added.
FDA and Citizen Petition Status
Asked about the status of the petition from Valisure, an FDA spokesperson said: “The FDA does not comment on the status of pending petitions.”
Dr. Barbieri and Dr. Lim had no relevant disclosures. There were no funding sources for either of the two studies.
A version of this article first appeared on Medscape.com.
Generational Differences in Isotretinoin Prescribing Habits: A Cross-Sectional Analysis
To the Editor:
Prescriptions for isotretinoin may be influenced by patient demographics, medical comorbidities, and drug safety programs.1,2 In 1982, isotretinoin was approved by the US Food and Drug Administration for treatment of severe recalcitrant nodulocystic acne that is nonresponsive to conventional therapies such as antibiotics; however, prescriber beliefs regarding the necessity of oral antibiotic failure before isotretinoin is prescribed may be influenced by the provider’s generational age.3 Currently, there is a knowledge gap regarding the impact of provider characteristics, including the year providers completed training, on isotretinoin utilization. The aim of our cross-sectional study was to characterize generational isotretinoin prescribing habits in a large-scale midwestern private practice dermatology group.
Modernizing Medicine (https://www.modmed.com), an electronic medical record software, was queried for all encounters that included both an International Classification of Diseases, Tenth Revision, Clinical Modification diagnosis code L70.0 (acne vulgaris) and a medication prescription from May 2021 to May 2022. Data were collected from a large private practice group with locations across the state of Ohio. Exclusion criteria included provider-patient prescription pairs that included non–acne medication prescriptions, patients seen by multiple providers, and providers who treated fewer than 5 patients with acne during the study period. A mixed-effect multiple logistic regression was performed to analyze whether a patient was ever prescribed isotretinoin, adjusting for individual prescriber, prescriber generation (millennial [1981–1996], Generation X [1965–1980], and baby boomer [1946–1964]),4 and patient sex; spironolactone and oral antibiotic prescriptions during the study period were included as additional covariates in a subsequent post hoc analysis. This study utilized data that was fully deidentified in accordance with the US Health Insurance Portability and Accountability Act (HIPAA) Privacy Rule. Approval from an institutional review board was not required.
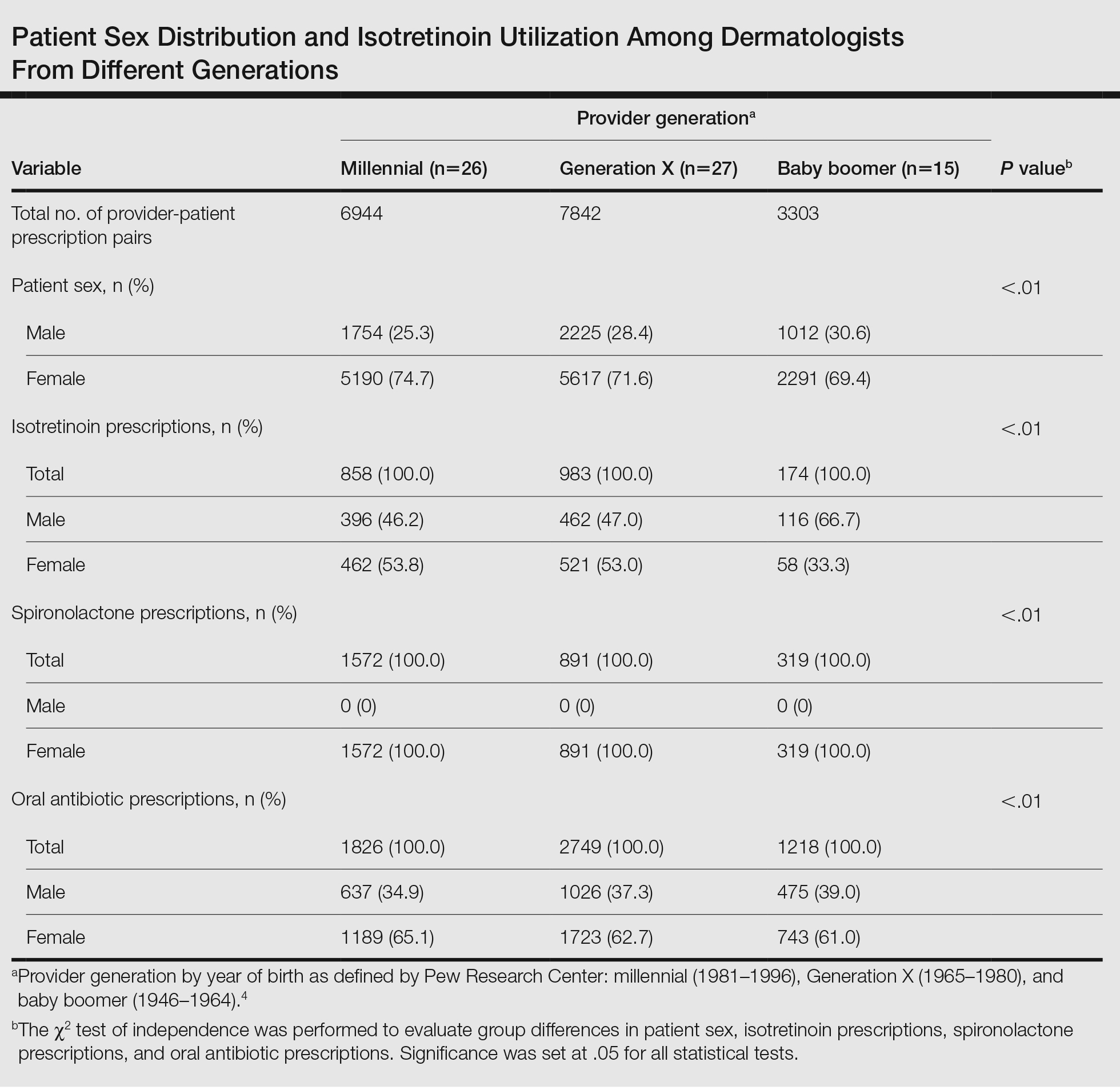
A total of 18,089 provider-patient prescription pairs were included in our analysis (Table). In our most robust model, female patients were significantly less likely to receive isotretinoin compared with male patients (adjusted OR [aOR], 0.394; P<.01). Millennial providers were significantly more likely to utilize isotretinoin in patients who did not receive antibiotics compared with patients who did receive antibiotics (aOR, 1.693; P<.01). When compared with both Generation X and baby boomers, millennial providers were more likely to prescribe isotretinoin in patients who received antibiotics (aOR, 2.227 [P=.02] and 3.638 [P<.01], respectively).
In 2018, the American Academy of Dermatology and the Global Alliance to Improve Outcomes in Acne updated thir guidelines to recommend isotretinoin as a first-line therapy for severe nodular acne, treatment-resistant moderate acne, or acne that produces scarring or psychosocial distress.5 Our study results suggest that millennial providers are adhering to these guidelines and readily prescribing isotretinoin in patients who did not receive antibiotics, which corroborates survey findings by Nagler and Orlow.3 Our results also revealed that prescriber generation may influence isotretinoin usage, with millennials utilizing isotretinoin more in patients who received oral antibiotic therapy than their older counterparts. In part, this may be due to beliefs among older generations that failure of oral antibiotics is necessary before pursuing isotretinoin.3 Additionally, this finding suggests that millennials, if utilizing antibiotics for acne, may have a lower threshold for starting isotretinoin in patients who received oral antibiotic therapy.
Generational prescribing variation appears not to be unique to isotretinoin and also may be present in the use of spironolactone. Over the past decade, utilization of spironolactone for acne treatment has increased, likely in response to new data demonstrating that routine use is safe and effective.6 Several large cohort and retrospective studies have debunked the historical concerns for tumorigenicity in those with breast cancer history as well as the need for routine laboratory monitoring for hyperkalemia.7,8 Although spironolactone use for the treatment of acne has increased, it still remains relatively underutilized,6 suggesting there may be a knowledge gap similar to that of isotretinoin, with younger generations utilizing spironolactone more readily than older generations.
Our study analyzed generational differences in isotretinoin utilization for acne over 1 calendar year. Limitations include sampling from a midwestern patient cohort and private practice–based providers. Due to limitations of our data set, we were unable to capture acne medication usage prior to May 2021, temporal sequencing of acne medication usage, and stratification of patients by acne severity. Furthermore, we were unable to capture female patients who were pregnant or planning pregnancy at the time of their encounter, which would exclude isotretinoin usage.
Overall, millennial providers may be utilizing isotretinoin more in line with the updated acne guidelines5 compared with providers from older generations. Further research is necessary to elucidate how these prescribing habits may change based on acne severity.
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity and sex with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Barbieri JS, Frieden IJ, Nagler AR. Isotretinoin, patient safety, and patient-centered care-time to reform iPLEDGE. JAMA Dermatol. 2020;156:21-22. doi:10.1001/jamadermatol.2019.3270
- Nagler AR, Orlow SJ. Dermatologists’ attitudes, prescription, and counseling patterns for isotretinoin: a questionnaire-based study. J Drugs Dermatol. 2015;14:184-189.
- Dimock M. Where Millennials end and Generation Z begins. Pew Research Center website. January 17, 2019. Accessed June 17, 2024. https://www.pewresearch.org/fact-tank/2019/01/17/where-millennials-end-and-generation-z-begins/
- Thiboutot DM, Dréno B, Abanmi A, et al. Practical management of acne for clinicians: an international consensus from the Global Alliance to Improve Outcomes in Acne. J Am Acad Dermatol. 2018;78(2 suppl 1):S1-S23.e1. doi:10.1016/j.jaad.2017.09.078
- Guzman AK, Barbieri JS. Comparative analysis of prescribing patterns of tetracycline class antibiotics and spironolactone between advanced practice providers and physicians in the treatment of acne vulgaris. J Am Acad Dermatol. 2021;84:1119-1121. doi:10.1016/j.jaad.2020.06.044
- Wei C, Bovonratwet P, Gu A, et al. Spironolactone use does not increase the risk of female breast cancer recurrence: a retrospective analysis. J Am Acad Dermatol. 2020;83:1021-1027. doi:10.1016/j.jaad.2020.05.081
- Plovanich M, Weng QY, Mostaghimi A. Low usefulness of potassium monitoring among healthy young women taking spironolactone for acne. JAMA Dermatol. 2015;151:941-944. doi:10.1001/jamadermatol.2015.34
To the Editor:
Prescriptions for isotretinoin may be influenced by patient demographics, medical comorbidities, and drug safety programs.1,2 In 1982, isotretinoin was approved by the US Food and Drug Administration for treatment of severe recalcitrant nodulocystic acne that is nonresponsive to conventional therapies such as antibiotics; however, prescriber beliefs regarding the necessity of oral antibiotic failure before isotretinoin is prescribed may be influenced by the provider’s generational age.3 Currently, there is a knowledge gap regarding the impact of provider characteristics, including the year providers completed training, on isotretinoin utilization. The aim of our cross-sectional study was to characterize generational isotretinoin prescribing habits in a large-scale midwestern private practice dermatology group.
Modernizing Medicine (https://www.modmed.com), an electronic medical record software, was queried for all encounters that included both an International Classification of Diseases, Tenth Revision, Clinical Modification diagnosis code L70.0 (acne vulgaris) and a medication prescription from May 2021 to May 2022. Data were collected from a large private practice group with locations across the state of Ohio. Exclusion criteria included provider-patient prescription pairs that included non–acne medication prescriptions, patients seen by multiple providers, and providers who treated fewer than 5 patients with acne during the study period. A mixed-effect multiple logistic regression was performed to analyze whether a patient was ever prescribed isotretinoin, adjusting for individual prescriber, prescriber generation (millennial [1981–1996], Generation X [1965–1980], and baby boomer [1946–1964]),4 and patient sex; spironolactone and oral antibiotic prescriptions during the study period were included as additional covariates in a subsequent post hoc analysis. This study utilized data that was fully deidentified in accordance with the US Health Insurance Portability and Accountability Act (HIPAA) Privacy Rule. Approval from an institutional review board was not required.
A total of 18,089 provider-patient prescription pairs were included in our analysis (Table). In our most robust model, female patients were significantly less likely to receive isotretinoin compared with male patients (adjusted OR [aOR], 0.394; P<.01). Millennial providers were significantly more likely to utilize isotretinoin in patients who did not receive antibiotics compared with patients who did receive antibiotics (aOR, 1.693; P<.01). When compared with both Generation X and baby boomers, millennial providers were more likely to prescribe isotretinoin in patients who received antibiotics (aOR, 2.227 [P=.02] and 3.638 [P<.01], respectively).

In 2018, the American Academy of Dermatology and the Global Alliance to Improve Outcomes in Acne updated thir guidelines to recommend isotretinoin as a first-line therapy for severe nodular acne, treatment-resistant moderate acne, or acne that produces scarring or psychosocial distress.5 Our study results suggest that millennial providers are adhering to these guidelines and readily prescribing isotretinoin in patients who did not receive antibiotics, which corroborates survey findings by Nagler and Orlow.3 Our results also revealed that prescriber generation may influence isotretinoin usage, with millennials utilizing isotretinoin more in patients who received oral antibiotic therapy than their older counterparts. In part, this may be due to beliefs among older generations that failure of oral antibiotics is necessary before pursuing isotretinoin.3 Additionally, this finding suggests that millennials, if utilizing antibiotics for acne, may have a lower threshold for starting isotretinoin in patients who received oral antibiotic therapy.
Generational prescribing variation appears not to be unique to isotretinoin and also may be present in the use of spironolactone. Over the past decade, utilization of spironolactone for acne treatment has increased, likely in response to new data demonstrating that routine use is safe and effective.6 Several large cohort and retrospective studies have debunked the historical concerns for tumorigenicity in those with breast cancer history as well as the need for routine laboratory monitoring for hyperkalemia.7,8 Although spironolactone use for the treatment of acne has increased, it still remains relatively underutilized,6 suggesting there may be a knowledge gap similar to that of isotretinoin, with younger generations utilizing spironolactone more readily than older generations.
Our study analyzed generational differences in isotretinoin utilization for acne over 1 calendar year. Limitations include sampling from a midwestern patient cohort and private practice–based providers. Due to limitations of our data set, we were unable to capture acne medication usage prior to May 2021, temporal sequencing of acne medication usage, and stratification of patients by acne severity. Furthermore, we were unable to capture female patients who were pregnant or planning pregnancy at the time of their encounter, which would exclude isotretinoin usage.
Overall, millennial providers may be utilizing isotretinoin more in line with the updated acne guidelines5 compared with providers from older generations. Further research is necessary to elucidate how these prescribing habits may change based on acne severity.
To the Editor:
Prescriptions for isotretinoin may be influenced by patient demographics, medical comorbidities, and drug safety programs.1,2 In 1982, isotretinoin was approved by the US Food and Drug Administration for treatment of severe recalcitrant nodulocystic acne that is nonresponsive to conventional therapies such as antibiotics; however, prescriber beliefs regarding the necessity of oral antibiotic failure before isotretinoin is prescribed may be influenced by the provider’s generational age.3 Currently, there is a knowledge gap regarding the impact of provider characteristics, including the year providers completed training, on isotretinoin utilization. The aim of our cross-sectional study was to characterize generational isotretinoin prescribing habits in a large-scale midwestern private practice dermatology group.
Modernizing Medicine (https://www.modmed.com), an electronic medical record software, was queried for all encounters that included both an International Classification of Diseases, Tenth Revision, Clinical Modification diagnosis code L70.0 (acne vulgaris) and a medication prescription from May 2021 to May 2022. Data were collected from a large private practice group with locations across the state of Ohio. Exclusion criteria included provider-patient prescription pairs that included non–acne medication prescriptions, patients seen by multiple providers, and providers who treated fewer than 5 patients with acne during the study period. A mixed-effect multiple logistic regression was performed to analyze whether a patient was ever prescribed isotretinoin, adjusting for individual prescriber, prescriber generation (millennial [1981–1996], Generation X [1965–1980], and baby boomer [1946–1964]),4 and patient sex; spironolactone and oral antibiotic prescriptions during the study period were included as additional covariates in a subsequent post hoc analysis. This study utilized data that was fully deidentified in accordance with the US Health Insurance Portability and Accountability Act (HIPAA) Privacy Rule. Approval from an institutional review board was not required.
A total of 18,089 provider-patient prescription pairs were included in our analysis (Table). In our most robust model, female patients were significantly less likely to receive isotretinoin compared with male patients (adjusted OR [aOR], 0.394; P<.01). Millennial providers were significantly more likely to utilize isotretinoin in patients who did not receive antibiotics compared with patients who did receive antibiotics (aOR, 1.693; P<.01). When compared with both Generation X and baby boomers, millennial providers were more likely to prescribe isotretinoin in patients who received antibiotics (aOR, 2.227 [P=.02] and 3.638 [P<.01], respectively).

In 2018, the American Academy of Dermatology and the Global Alliance to Improve Outcomes in Acne updated thir guidelines to recommend isotretinoin as a first-line therapy for severe nodular acne, treatment-resistant moderate acne, or acne that produces scarring or psychosocial distress.5 Our study results suggest that millennial providers are adhering to these guidelines and readily prescribing isotretinoin in patients who did not receive antibiotics, which corroborates survey findings by Nagler and Orlow.3 Our results also revealed that prescriber generation may influence isotretinoin usage, with millennials utilizing isotretinoin more in patients who received oral antibiotic therapy than their older counterparts. In part, this may be due to beliefs among older generations that failure of oral antibiotics is necessary before pursuing isotretinoin.3 Additionally, this finding suggests that millennials, if utilizing antibiotics for acne, may have a lower threshold for starting isotretinoin in patients who received oral antibiotic therapy.
Generational prescribing variation appears not to be unique to isotretinoin and also may be present in the use of spironolactone. Over the past decade, utilization of spironolactone for acne treatment has increased, likely in response to new data demonstrating that routine use is safe and effective.6 Several large cohort and retrospective studies have debunked the historical concerns for tumorigenicity in those with breast cancer history as well as the need for routine laboratory monitoring for hyperkalemia.7,8 Although spironolactone use for the treatment of acne has increased, it still remains relatively underutilized,6 suggesting there may be a knowledge gap similar to that of isotretinoin, with younger generations utilizing spironolactone more readily than older generations.
Our study analyzed generational differences in isotretinoin utilization for acne over 1 calendar year. Limitations include sampling from a midwestern patient cohort and private practice–based providers. Due to limitations of our data set, we were unable to capture acne medication usage prior to May 2021, temporal sequencing of acne medication usage, and stratification of patients by acne severity. Furthermore, we were unable to capture female patients who were pregnant or planning pregnancy at the time of their encounter, which would exclude isotretinoin usage.
Overall, millennial providers may be utilizing isotretinoin more in line with the updated acne guidelines5 compared with providers from older generations. Further research is necessary to elucidate how these prescribing habits may change based on acne severity.
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity and sex with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Barbieri JS, Frieden IJ, Nagler AR. Isotretinoin, patient safety, and patient-centered care-time to reform iPLEDGE. JAMA Dermatol. 2020;156:21-22. doi:10.1001/jamadermatol.2019.3270
- Nagler AR, Orlow SJ. Dermatologists’ attitudes, prescription, and counseling patterns for isotretinoin: a questionnaire-based study. J Drugs Dermatol. 2015;14:184-189.
- Dimock M. Where Millennials end and Generation Z begins. Pew Research Center website. January 17, 2019. Accessed June 17, 2024. https://www.pewresearch.org/fact-tank/2019/01/17/where-millennials-end-and-generation-z-begins/
- Thiboutot DM, Dréno B, Abanmi A, et al. Practical management of acne for clinicians: an international consensus from the Global Alliance to Improve Outcomes in Acne. J Am Acad Dermatol. 2018;78(2 suppl 1):S1-S23.e1. doi:10.1016/j.jaad.2017.09.078
- Guzman AK, Barbieri JS. Comparative analysis of prescribing patterns of tetracycline class antibiotics and spironolactone between advanced practice providers and physicians in the treatment of acne vulgaris. J Am Acad Dermatol. 2021;84:1119-1121. doi:10.1016/j.jaad.2020.06.044
- Wei C, Bovonratwet P, Gu A, et al. Spironolactone use does not increase the risk of female breast cancer recurrence: a retrospective analysis. J Am Acad Dermatol. 2020;83:1021-1027. doi:10.1016/j.jaad.2020.05.081
- Plovanich M, Weng QY, Mostaghimi A. Low usefulness of potassium monitoring among healthy young women taking spironolactone for acne. JAMA Dermatol. 2015;151:941-944. doi:10.1001/jamadermatol.2015.34
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity and sex with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Barbieri JS, Frieden IJ, Nagler AR. Isotretinoin, patient safety, and patient-centered care-time to reform iPLEDGE. JAMA Dermatol. 2020;156:21-22. doi:10.1001/jamadermatol.2019.3270
- Nagler AR, Orlow SJ. Dermatologists’ attitudes, prescription, and counseling patterns for isotretinoin: a questionnaire-based study. J Drugs Dermatol. 2015;14:184-189.
- Dimock M. Where Millennials end and Generation Z begins. Pew Research Center website. January 17, 2019. Accessed June 17, 2024. https://www.pewresearch.org/fact-tank/2019/01/17/where-millennials-end-and-generation-z-begins/
- Thiboutot DM, Dréno B, Abanmi A, et al. Practical management of acne for clinicians: an international consensus from the Global Alliance to Improve Outcomes in Acne. J Am Acad Dermatol. 2018;78(2 suppl 1):S1-S23.e1. doi:10.1016/j.jaad.2017.09.078
- Guzman AK, Barbieri JS. Comparative analysis of prescribing patterns of tetracycline class antibiotics and spironolactone between advanced practice providers and physicians in the treatment of acne vulgaris. J Am Acad Dermatol. 2021;84:1119-1121. doi:10.1016/j.jaad.2020.06.044
- Wei C, Bovonratwet P, Gu A, et al. Spironolactone use does not increase the risk of female breast cancer recurrence: a retrospective analysis. J Am Acad Dermatol. 2020;83:1021-1027. doi:10.1016/j.jaad.2020.05.081
- Plovanich M, Weng QY, Mostaghimi A. Low usefulness of potassium monitoring among healthy young women taking spironolactone for acne. JAMA Dermatol. 2015;151:941-944. doi:10.1001/jamadermatol.2015.34
Practice Points
- Provider generational age appears to impact utilization of isotretinoin for the treatment of acne.
- Millennial providers seem to adhere more readily to guidelines for precribing isotretinoin vs older generations and also may have a lower threshold for starting isotretinoin in patients who received oral antibiotic therapy for acne treatment.
Transgender and Gender Diverse Health Care in the US Military: What Dermatologists Need to Know
People whose gender identity differs from the sex assigned at birth are referred to as transgender. For some, gender identity may not fit into the binary constructs of male and female but rather falls between, within, or outside this construct. These people often consider themselves nonbinary or gender diverse. As the terminology continues to evolve, current recommendations include referring to this patient population as transgender and gender diverse (TGD) to ensure the broadest inclusivity.1 In this article, the following terms are used as defined below:
- The terms transgender woman and trans feminine describe persons who were assigned male gender at birth but their affirmed gender is female or nonmasculine.
- The terms transgender man and trans masculine describe persons who were assigned female gender at birth but their affirmed gender is male or nonfeminine.
The US Military’s policies on the service of TGD persons have evolved considerably over the past decade. Initial military policies barred TGD service members (TSMs) from service all together, leading to challenges in accessing necessary health care. The first official memorandum explicitly allowing military service by TGD persons was released on June 30, 2016.2 The intention of this memorandum was 2-fold: (1) to allow TGD persons to serve in the military so long as they meet “the rigorous standards for military service and readiness” by fulfilling the same standards and procedures as other military service members, including medical fitness for duty, physical fitness, uniform and grooming, deployability, and retention, and (2) to direct the establishment of new or updated policies to specific departments and prescribe procedures for retention standards, separation from service, in-service transition, and medical coverage.2 Several other official policies were released following this initial memorandum that provided more specific guidance on how to implement these policies at the level of the force, unit, and individual service member.
Modifications to the original 2016 policies had varying impacts on transgender health care provision and access.3 At the time of publication of this article, the current policy—the Department of Defense Instruction 1300.284—among others, establishes standards and procedures for the process by which active and reserve TSMs may medically, socially, and legally transition genders within the military. The current policy applies to all military branches and serves as the framework by which each branch currently organizes their gender-affirmation processes (GAP).4
There currently are several different GAP models among the military branches.5 Each branch has a different model or approach to implementing the current policy, with varying service-specific processes in place for TSMs to access gender-affirming care; however, this may be changing. The Defense Health Agency is in the process of consolidating and streamlining the GAP across the Department of Defense branches in an effort to optimize costs and ensure uniformity of care. Per the Defense Health Agency Procedural Instruction Number 6025.21 published in May 2023, the proposed consolidated model likely will entail a single central transgender health center that provides oversight and guidance for several regional joint-service gender-affirming medical hubs. Patients would either be managed at the level of the hub or be referred to the central site.5
Herein, we discuss the importance of gender-affirming care and how military and civilian dermatologists can contribute. We also review disparities in health care and identify areas of improvement.
Benefits of Gender-Affirming Care
Gender-affirming procedures are critical for aligning physical appearance with gender identity. Physical appearance is essential for psychological well-being, operational readiness, and the safety of TSMs.6 It is well documented that TGD persons experience suicidal ideation, depression, stigma, discrimination and violence at higher rates than their cisgender peers.7,8 It is important to recognize that transgender identity is not a mental illness, and these elevated rates have been linked to complex trauma, societal stigma, violence, and discrimination.1 Other studies have suggested that increased access to gender-affirming interventions may ameliorate these mental health concerns.1,7-9
The major components of gender-affirming care include hormone therapy, gender confirmation surgery, and mental health care, if needed. These are covered by TRICARE, the health care program for military service members; however, at the time of publication, many of the dermatologic gender-affirming procedures are not covered by TRICARE because they are considered “cosmetic procedures,” which is a term used by insurance companies but does not accurately indicate whether a procedure is medically necessary or not. Newer literature has demonstrated that gender-affirming care positively affects the lives of TGD patients, strengthening the argument that gender-affirming care is a medical necessity and not just cosmetic.1
Aesthetic Procedures in Gender-Affirming Care
Surgeons, including those within the specialties of oto-laryngology, oral and maxillofacial surgery, urology, gynecology, and plastic surgery, provide major gender-affirming interventions; however, dermatologists may offer less invasive solutions that can serve as a temporary experience prior to undergoing more permanent procedures.Hormonally driven disorders including acne, hair loss, and melasma also are managed by dermatologists, along with scar treatment following surgeries.
Because human variation is expansive and subjective, what is considered feminine or masculine may vary by person, group, culture, and country; therefore, it is imperative to ask patients about their individual aesthetic goals and tailor their treatment accordingly. Feminine and masculine are terms that will be used to describe prototypical appearances and are not meant to define a patient’s current state or ultimate goals. The following procedures and medical interventions are where dermatologists can play an important role in TGD persons’ GAPs.
Botulinum Toxin Injections—Botulinum toxin injection is the most common nonsurgical aesthetic procedure performed around the world.10 The selective paralysis afforded by botulinum toxin has several uses for people undergoing transition. Aesthetically, the feminine eyebrow tends to be positioned above the orbital rim and is arched with its apex between the lateral limbus and lateral canthus,11 while the masculine eyebrow tends to be flatter and fuller and runs over the orbital rim without a peak. For people seeking a more feminine appearance, an eyebrow lift with botulinum toxin can help reshape the typical flatter masculine eyebrow to give it lateral lift that often is considered more feminine. The targeted muscle is the superolateral orbicularis oculi, which serves as a depressor on the eyebrow. This can be combined with purposefully avoiding total lateral frontalis paralysis, which leads to a “Spock” brow for extra lift. Conversely, a naturally arched and higher eyebrow can be flattened and lowered by selectively targeting areas of the frontalis muscle.
Broad square jawlines typically are considered a masculine feature and are another area where botulinum toxin can be used to feminize a patient’s facial features. Targeting the masseter muscle induces muscle weakness, which ultimately may result in atrophy after one or more treatment sessions. This atrophy may lead to narrowing of the lower face and thus may lead to a fuller-appearing midface or overall more heart-shaped face. Every individual’s aesthetic goals are unique and therefore should be discussed prior to any treatment.
Dermal Fillers—Dermal fillers are gel-like substances injected under the skin for subtle contouring of the face. Fillers also can be used to help promote a more masculine or feminine appearance. Filler can be placed in the lips to create a fuller, more projected, feminine-appearing lip. Malar cheek and central lower chin filler can be used to help define a heart-shaped face by accentuating the upper portion of the face and creating a more pointed chin, respectively. Alternatively, filler can be used to masculinize the chin by placing it where it can increase jawline squareness and increase anterior jaw projection. Additionally, filler at the angle of the jaw can help accentuate a square facial shape and a more defined jawline. Although not as widely practiced, lateral brow filler can create a heavier-appearing and broader forehead for a more masculine appearance. These procedures can be combined with the previously mentioned botulinum toxin procedures for a synergistic effect.
Deoxycholic Acid—Deoxycholic acid is an injectable product used to selectively remove unwanted fat. It currently is approved by the US Food and Drug Administration for submental fat, but some providers are experimenting with off-label uses. Buccal fat pad removal—or in this case reduction by dissolution—tends to give a thinner, more feminine facial appearance.12 Reducing fat around the axillae also can help promote a more masculine upper torso.13 The safety of deoxycholic acid in these areas has not been adequately tested; thus, caution should be used when discussing these off-label uses with patients.
Hair and Tattoo Removal—Hair removal may be desired by TGD persons for a variety of reasons. Because cisgender females tend to have less body hair overall, transgender people in pursuit of a more feminine appearance often desire removal of facial, neck, and body hair. Although shaving and other modalities such as waxing and chemical depilatories are readily available at-home options, they are not permanent and may lead to folliculitis or pseudofolliculitis barbae. Laser hair removal (LHR) and electrolysis are modalities provided by dermatologists that tend to be more permanent and lead to better outcomes, including less irritation and better aesthetic appearance. It is important to keep in mind that not every person and not every body site can be safely treated with LHR. Patients with lighter skin types and darker hair tend to have the most effective response with a higher margin of safety, as these features allow the laser energy to be selectively absorbed by the melanin in the hair bulb and not by the background skin pigmentation.14,15 Inappropriate patient selection or improper settings for wavelength, pulse width, or fluences can lead to burns and permanent scarring.14,15 Electrolysis is an alternative to hair removal within tattoos and is more effective for those individuals with blonde, red, or white hair.16
Another novel treatment for unwanted hair is eflornithine hydrochloride cream, which works by blocking ornithine decarboxylase, the enzyme that stimulates hair growth. It currently is approved to reduce unwanted hair on the face and adjacent areas under the chin; however the effects of this medication are modest and the medication can be expensive.17
Cosmetic hair and tattoo removal are not currently covered by TRICARE, except in cases of surgical and donor-site preparation for some GAPs. Individuals may desire removal of tattoos at surgery sites to obtain more natural-appearing skin. Currently, GAPs such as vaginoplasty, phalloplasty, and metoidioplasty—often referred to by patients as “bottom surgeries”—include insurance coverage for tattoo removal, LHR, and/or electrolysis.
Management of Hormonal Adverse Effects
Acne—Individuals on testosterone supplementation tend to develop acne for the first several years of treatment, but it may improve with time.18 Acne is treated in individuals receiving testosterone the same way as it is treated in cisgender men, with numerous options for topical and oral medications. In trans masculine persons, spironolactone therapy typically is avoided because it may interfere with the actions of exogenous testosterone administered as part of gender-affirming medical treatment and may lead to other undesired adverse effects such as impotence and gynecomastia.1
Although acne typically improves after starting estrogen therapy, patients receiving estrogens may still develop acne. Most trans feminine patients will already be on an estrogen and an antiandrogen, often spironolactone.1 Spironolactone often is used as monotherapy for acne control in cisgender women. Additionally, an important factor to consider with spironolactone is the possible adverse effect of increased micturition. Currently, the military rarely has gender-inclusive restroom options, which can create a challenge for TSMs who find themselves needing to use the restroom more frequently in the workplace.
If planning therapy with isotretinoin, dermatologists should discuss several important factors with all patients, including TGD patients. One consideration is the patient’s planned future surgeries. Although new literature shows that isotretinoin does not adversely affect wound healing,19 some surgeons still adhere to an isotretinoin washout period of 6 to 12 months prior to performing any elective procedures due to concerns about wound healing.20,21 Second, be sure to properly assess and document pregnancy potential in TGD persons. Providers should not assume that a patient is not pregnant or is not trying to become pregnant just because they are trans masculine. It also is important to note that testosterone is not a reliable birth control method.1 If a patient still has ovaries, fallopian tubes, and a uterus, they are considered medically capable of pregnancy, and providers should keep this in mind regarding all procedures in the TGD population.
Another newer acne treatment modality is the 1762-nm laser, which targets sebaceous glands.22 This device allows for targeted treatment of acne-prone areas without systemic therapy such as retinoids or antiandrogens. The 1762-nm laser is not widely available but may become a regular treatment option once its benefits are proven over time.
Alopecia and Hyperpigmentation—Androgens, whether endogenously or exogenously derived, can lead to androgenetic alopecia (AGA) in genetically susceptible individuals. Trans masculine persons and others receiving androgen therapy are at higher risk for AGA, which often is undesirable and may be considered gender affirming by some TGD persons. Standard AGA treatments for cisgender men also can be used in trans masculine persons. Some of the most common anti-AGA medications are topical minoxidil, oral finasteride, and oral minoxidil. Although Coleman et al1 recently reported that finasteride may be an appropriate treatment option in trans masculine persons experiencing alopecia, treatment with 5α-reductase inhibitors may impair clitoral growth and the development of facial and body hair. Further studies are needed to assess the efficacy and safety of 5α-reductase inhibitors in transgender populations.1 Dutasteride may be used off-label and comes with a similar potential adverse-event profile as finasteride, which includes depression, decreased libido, erectile dysfunction, ejaculation disorders, and gynecomastia.
Conversely, AGA tends to improve in trans feminine persons and others receiving estrogen and antiandrogen therapy. Natural testosterone production is suppressed by estrogens and spironolactone as well as in patients who undergo orchiectomy.1 Although spironolactone is not approved for acne, AGA, or hirsutism, it is a standard treatment of AGA in cisgender women because it functions to block the effects of androgens, including at the hair follicle. Finasteride may be used for AGA in cisgender women but it is not recommended for trans feminine persons.1
There are many other modalities available for the treatment of AGA that are less commonly used—some may be cost prohibitive or do not have robust supporting evidence, or both. One example is hair
Melasma is a hyperpigmentation disorder related to estrogens, UV light exposure, and sometimes medication use (eg, hormonal birth control, spironolactone).24 The mainstay of treatment is prevention, including sun avoidance as well as use of sun-protective clothing and broad-spectrum sunscreens. Dermatologists tend to recommend physical sunscreens containing zinc oxide, titanium dioxide, and/or iron oxide, as they cover a wider UV spectrum and also provide some protection from visible light. Once melasma is present, dermatologists still have several treatment options. Topical hydroquinone is a proven treatment; however, it must be used with caution to avoid ochronosis. With careful patient selection, chemical peels also are effective treatment options for dyspigmentation and hyperpigmentation. Energy devices such as intense pulsed light and tattoo removal lasers—Q-switched lasers and picosecond pulse widths—also can be used to treat hyperpigmentation. Oral, intralesional, and topical tranexamic acid are newer treatment options for melasma that still are being studied and have shown promising results. Further studies are needed to determine long-term safety and optimal treatment regimens.24,25
Many insurance carriers, including TRICARE, do not routinely cover medical management of AGA or melasma. Patients should be advised that they likely will have to pay for any medications prescribed and procedures undertaken for these purposes; however, some medication costs can be offset by ordering larger prescription quantities, such as a 90-day supply vs a 30-day supply, as well as utilizing pharmacy discount programs.
Scar Management Following Surgery
In TSMs who undergo gender-affirming surgeries, dermatologists play an important role when scar symptoms develop, including pruritus, tenderness, and/or paresthesia. In the military, some common treatment modalities for symptomatic scars include intralesional steroids with or without 5-fluouroruacil and the fractionated CO2 laser. There also are numerous experimental treatment options for scars, including intralesional or perilesional botulinum toxin, the pulsed dye laser, or nonablative fractionated lasers. These modalities also may be used on hypertrophic scars or keloids. Another option for keloids is scar excision followed by superficial radiation therapy.26
Mental Health Considerations
Providers must take psychological adverse effects into consideration when considering medical therapies for dermatologic conditions in TGD patients. In particular, it is important to consider the risks for increased rates of depression and suicidal ideation formerly associated with the use of isotretinoin and finasteride, though much of the evidence regarding these risks has been called into question in recent years.27,28 Nonetheless, it remains prominent in lay media and may be a more important consideration in patients at higher baseline risk.27 Although there are no known studies that have expressly assessed rates of depression or suicidal ideation in TGD patients taking isotretinoin or finasteride, it is well established that TGD persons are at higher baseline risk for depression and suicidality.1,7,8 All patients should be carefully assessed for depression and suicidal ideation as well as counseled regarding these risks prior to initiating these therapies. If concerns for untreated mental health issues arise during screening and counseling, patients should be referred for assessment by a behavioral health specialist prior to starting therapy.
Future Directions
The future of TGD health care in the military could see an expansion of covered benefits and the development of new dermatologic procedures or medications. Research and policy evolution are necessary to bridge the current gaps in care; however, it is unlikely that all procedures currently considered to be cosmetic will become covered benefits.
Facial LHR is a promising candidate for future coverage for trans feminine persons. When cisgender men develop adverse effects from mandatory daily shaving, LHR is already a covered benefit. Two arguments in support of adding LHR for TGD patients revolve around achieving and maintaining an appearance congruent with their gender along with avoiding unwanted adverse effects related to daily shaving. Visual conformity with one’s affirmed gender has been associated with improvements in well-being, quality of life, and some mental health conditions.29
Scar prevention, treatment, and reduction are additional areas under active research in which dermatologists likely will play a crucial role.30,31 As more dermatologic procedures are performed on TGD persons, the published data and collective knowledge regarding best practices in this population will continue to grow, which will lead to improved cosmetic and safety outcomes.
Final Thoughts
Although dermatologists do not directly perform gender-affirming surgeries or hormone management, they do play an important role in enhancing a TGD person’s desired appearance and managing possible adverse effects resulting from gender-affirming interventions. There have been considerable advancements in TGD health care over the past decade, but there likely are more changes on the way. As policies and understanding of TGD health care needs evolve, it is crucial that the military health care system adapts to provide comprehensive, accessible, and equitable care, which includes expanding the range of covered dermatologic treatments to fully support the health and readiness of TSMs.
Acknowledgment—We would like to extend our sincere appreciation to the invaluable contributions and editorial support provided by Allison Higgins, JD (San Antonio, Texas), throughout the writing of this article.
- Coleman E, Radix AE, Bouman WP, et al. Standards of care for the health of transgender and gender diverse people, version 8. Int J Transgend Health. 2022;23(suppl 1):S1-S260. doi:10.1080/26895269.2022.2100644
- Secretary of Defense. DTM 16-005—military service of transgender service members. June 30, 2016. Accessed June 17, 2024. https://dod.defense.gov/Portals/1/features/2016/0616_policy/DTM-16-005.pdf
- Office of the Deputy Secretary of Defense. DTM 19-004—military service by transgender persons and persons with gender dysphoria. March 17, 2020. Accessed June 17, 2024. https://health.mil/Reference-Center/Policies/2020/03/17/Military-Service-by-Transgender-Persons-and-Persons-with-Gender-Dysphoria
- Office of the Under Secretary of Defense for Personnel and Readiness. Department of Defense Instruction (DODI) 1300.28. in-service transition for transgender service members. September 4, 2020. Accessed June 17, 2024. https://health.mil/Reference-Center/Policies/2020/09/04/Military-Service-by-Transgender-Persons-and-Persons-with-Gender-Dysphoria
- Defense Health Agency Procedural Instruction Number 6025.21, Guidance for Gender-Affirming Health Care of Transgender and Gender-Diverse Active and Reserve Component Service Members, May 12, 2023. https://www.health.mil/Reference-Center/DHA-Publications/2023/05/12/DHA-PI-6015-21
- Elders MJ, Brown GR, Coleman E, et al. Medical aspects of transgender military service. Armed Forces Soc. 2015;41:199-220. doi:10.1177/0095327X14545625.
- Almazan AN, Keuroghlian AS. Association between gender-affirming surgeries and mental health outcomes. JAMA Surg. 2021;156:611-618.
- Tordoff DM, Wanta JW, Collin A, et al. Mental health outcomes in transgender and nonbinary youths receiving gender-affirming care. JAMA Netw Open. 2022;5:E220978. doi:10.1001/jamanetworkopen.2022.0978
- Olson-Kennedy J, Warus J, Okonta V, et al. Chest reconstruction and chest dysphoria in transmasculine minors and young adults: comparisons of nonsurgical and postsurgical cohorts. JAMA Pediatr. 2018;172:431-436. doi:10.1001/jamapediatrics.2017.5440
- Top non-invasive cosmetic procedures worldwide 2022. Statista website. February 8, 2024. Accessed June 13, 2024. https://www.statista.com/statistics/293449/leading-nonsurgical-cosmetic-procedures/
- Kashkouli MB, Abdolalizadeh P, Abolfathzadeh N, et al. Periorbital facial rejuvenation; applied anatomy and pre-operative assessment. J Curr Ophthalmol. 2017;29:154-168. doi:10.1016/j.joco.2017.04.001
- Thomas MK, D’Silva JA, Borole AJ. Injection lipolysis: a systematic review of literature and our experience with a combination of phosphatidylcholine and deoxycholate over a period of 14 years in 1269 patients of Indian and South East Asian origin. J Cutan Aesthet Surg. 2018;11:222-228. doi:10.4103/JCAS.JCAS_117_18
- Jegasothy SM. Deoxycholic acid injections for bra-line lipolysis. Dermatol Surg. 2018;44:757-760. doi:10.1097/DSS.0000000000001311
- Dierickx CC. Hair removal by lasers and intense pulsed light sources. Dermatol Clin. 2002;20:135-146. doi:10.1016/s0733-8635(03)00052-4
- Lepselter J, Elman M. Biological and clinical aspects in laser hair removal. J Dermatolog Treat. 2004;15:72-83. doi:10.1080/09546630310023152
- Yuan N, Feldman AT, Chin P, et al. Comparison of permanent hair removal procedures before gender-affirming vaginoplasty: why we should consider laser hair removal as a first-line treatment for patients who meet criteria. Sex Med. 2022;10:100545. doi:10.1016/j.esxm.2022.100545
- Kumar A, Naguib YW, Shi YC, et al. A method to improve the efficacy of topical eflornithine hydrochloride cream. Drug Deliv. 2016;23:1495-1501. doi:10.3109/10717544.2014.951746
- Hembree WC, Cohen-Kettenis PT, Gooren L, et al. Endocrine treatment of gender-dysphoric/gender-incongruent persons: an endocrine society clinical practice guideline. J Clin Endocrinol Metabol. 2017;102:3869-3903.
- Hatami P, Balighi K, Asl HN, et al. Isotretinoin and timing of procedural interventions: clinical implications and practical points. J Cosmet Dermatol. 2023;22:2146-2149. doi:10.1111/jocd.15874
- Rubenstein R, Roenigk HH Jr, Stegman SJ, et al. Atypical keloids after dermabrasion of patients taking isotretinoin. J Am Acad Dermatol. 1986;15(2 pt 1):280-285.
- Zachariae H. Delayed wound healing and keloid formation following argon laser treatment or dermabrasion during isotretinoin treatment. Br J Dermatol. 1988;118:703-706.
- Goldberg D, Kothare A, Doucette M, et al. Selective photothermolysis with a novel 1726 nm laser beam: a safe and effective solution for acne vulgaris. J Cosmet Dermatol. 2023;22:486-496. doi:10.1111/jocd.15602
- Sun HY, Sebaratnam DF. Clascoterone as a novel treatment for androgenetic alopecia. Clin Exp Dermatol. 2020;45:913-914. doi:10.1111/ced.14292
- Bolognia JL, Schaffer JV, Cerroni L. Dermatology: 2-Volume Set. Elsevier; 2024:1130.
- Konisky H, Balazic E, Jaller JA, et al. Tranexamic acid in melasma: a focused review on drug administration routes. J Cosmet Dermatol. 2023;22:1197-1206. doi:10.1111/jocd.15589
- Walsh LA, Wu E, Pontes D, et al. Keloid treatments: an evidence-based systematic review of recent advances. Syst Rev. 2023;12:42. doi:10.1186/s13643-023-02192-7
- Kridin K, Ludwig RJ. Isotretinoin and the risk of psychiatric disturbances: a global study shedding new light on a debatable story. J Am Acad Dermatol. 2023;88:388-394. doi:10.1016/j.jaad.2022.10.031
- Dyson TE, Cantrell MA, Lund BC. Lack of association between 5α-reductase inhibitors and depression. J Urol. 2020;204:793-798. doi:10.1097/JU.0000000000001079
- To M, Zhang Q, Bradlyn A, et al. Visual conformity with affirmed gender or “passing”: its distribution and association with depression and anxiety in a cohort of transgender people. J Sex Med. 2020;17:2084-2092. doi:10.1016/j.jsxm.2020.07.019
- Fernandes MG, da Silva LP, Cerqueira MT, et al. Mechanomodulatory biomaterials prospects in scar prevention and treatment. Acta Biomater. 2022;150:22-33. doi:10.1016/j.actbio.2022.07.042
- Kolli H, Moy RL. Prevention of scarring with intraoperative erbium:YAG laser treatment. J Drugs Dermatol. 2020;19:1040-1043. doi:10.36849/JDD.2020.5244
People whose gender identity differs from the sex assigned at birth are referred to as transgender. For some, gender identity may not fit into the binary constructs of male and female but rather falls between, within, or outside this construct. These people often consider themselves nonbinary or gender diverse. As the terminology continues to evolve, current recommendations include referring to this patient population as transgender and gender diverse (TGD) to ensure the broadest inclusivity.1 In this article, the following terms are used as defined below:
- The terms transgender woman and trans feminine describe persons who were assigned male gender at birth but their affirmed gender is female or nonmasculine.
- The terms transgender man and trans masculine describe persons who were assigned female gender at birth but their affirmed gender is male or nonfeminine.
The US Military’s policies on the service of TGD persons have evolved considerably over the past decade. Initial military policies barred TGD service members (TSMs) from service all together, leading to challenges in accessing necessary health care. The first official memorandum explicitly allowing military service by TGD persons was released on June 30, 2016.2 The intention of this memorandum was 2-fold: (1) to allow TGD persons to serve in the military so long as they meet “the rigorous standards for military service and readiness” by fulfilling the same standards and procedures as other military service members, including medical fitness for duty, physical fitness, uniform and grooming, deployability, and retention, and (2) to direct the establishment of new or updated policies to specific departments and prescribe procedures for retention standards, separation from service, in-service transition, and medical coverage.2 Several other official policies were released following this initial memorandum that provided more specific guidance on how to implement these policies at the level of the force, unit, and individual service member.
Modifications to the original 2016 policies had varying impacts on transgender health care provision and access.3 At the time of publication of this article, the current policy—the Department of Defense Instruction 1300.284—among others, establishes standards and procedures for the process by which active and reserve TSMs may medically, socially, and legally transition genders within the military. The current policy applies to all military branches and serves as the framework by which each branch currently organizes their gender-affirmation processes (GAP).4
There currently are several different GAP models among the military branches.5 Each branch has a different model or approach to implementing the current policy, with varying service-specific processes in place for TSMs to access gender-affirming care; however, this may be changing. The Defense Health Agency is in the process of consolidating and streamlining the GAP across the Department of Defense branches in an effort to optimize costs and ensure uniformity of care. Per the Defense Health Agency Procedural Instruction Number 6025.21 published in May 2023, the proposed consolidated model likely will entail a single central transgender health center that provides oversight and guidance for several regional joint-service gender-affirming medical hubs. Patients would either be managed at the level of the hub or be referred to the central site.5
Herein, we discuss the importance of gender-affirming care and how military and civilian dermatologists can contribute. We also review disparities in health care and identify areas of improvement.
Benefits of Gender-Affirming Care
Gender-affirming procedures are critical for aligning physical appearance with gender identity. Physical appearance is essential for psychological well-being, operational readiness, and the safety of TSMs.6 It is well documented that TGD persons experience suicidal ideation, depression, stigma, discrimination and violence at higher rates than their cisgender peers.7,8 It is important to recognize that transgender identity is not a mental illness, and these elevated rates have been linked to complex trauma, societal stigma, violence, and discrimination.1 Other studies have suggested that increased access to gender-affirming interventions may ameliorate these mental health concerns.1,7-9
The major components of gender-affirming care include hormone therapy, gender confirmation surgery, and mental health care, if needed. These are covered by TRICARE, the health care program for military service members; however, at the time of publication, many of the dermatologic gender-affirming procedures are not covered by TRICARE because they are considered “cosmetic procedures,” which is a term used by insurance companies but does not accurately indicate whether a procedure is medically necessary or not. Newer literature has demonstrated that gender-affirming care positively affects the lives of TGD patients, strengthening the argument that gender-affirming care is a medical necessity and not just cosmetic.1
Aesthetic Procedures in Gender-Affirming Care
Surgeons, including those within the specialties of oto-laryngology, oral and maxillofacial surgery, urology, gynecology, and plastic surgery, provide major gender-affirming interventions; however, dermatologists may offer less invasive solutions that can serve as a temporary experience prior to undergoing more permanent procedures.Hormonally driven disorders including acne, hair loss, and melasma also are managed by dermatologists, along with scar treatment following surgeries.
Because human variation is expansive and subjective, what is considered feminine or masculine may vary by person, group, culture, and country; therefore, it is imperative to ask patients about their individual aesthetic goals and tailor their treatment accordingly. Feminine and masculine are terms that will be used to describe prototypical appearances and are not meant to define a patient’s current state or ultimate goals. The following procedures and medical interventions are where dermatologists can play an important role in TGD persons’ GAPs.
Botulinum Toxin Injections—Botulinum toxin injection is the most common nonsurgical aesthetic procedure performed around the world.10 The selective paralysis afforded by botulinum toxin has several uses for people undergoing transition. Aesthetically, the feminine eyebrow tends to be positioned above the orbital rim and is arched with its apex between the lateral limbus and lateral canthus,11 while the masculine eyebrow tends to be flatter and fuller and runs over the orbital rim without a peak. For people seeking a more feminine appearance, an eyebrow lift with botulinum toxin can help reshape the typical flatter masculine eyebrow to give it lateral lift that often is considered more feminine. The targeted muscle is the superolateral orbicularis oculi, which serves as a depressor on the eyebrow. This can be combined with purposefully avoiding total lateral frontalis paralysis, which leads to a “Spock” brow for extra lift. Conversely, a naturally arched and higher eyebrow can be flattened and lowered by selectively targeting areas of the frontalis muscle.
Broad square jawlines typically are considered a masculine feature and are another area where botulinum toxin can be used to feminize a patient’s facial features. Targeting the masseter muscle induces muscle weakness, which ultimately may result in atrophy after one or more treatment sessions. This atrophy may lead to narrowing of the lower face and thus may lead to a fuller-appearing midface or overall more heart-shaped face. Every individual’s aesthetic goals are unique and therefore should be discussed prior to any treatment.
Dermal Fillers—Dermal fillers are gel-like substances injected under the skin for subtle contouring of the face. Fillers also can be used to help promote a more masculine or feminine appearance. Filler can be placed in the lips to create a fuller, more projected, feminine-appearing lip. Malar cheek and central lower chin filler can be used to help define a heart-shaped face by accentuating the upper portion of the face and creating a more pointed chin, respectively. Alternatively, filler can be used to masculinize the chin by placing it where it can increase jawline squareness and increase anterior jaw projection. Additionally, filler at the angle of the jaw can help accentuate a square facial shape and a more defined jawline. Although not as widely practiced, lateral brow filler can create a heavier-appearing and broader forehead for a more masculine appearance. These procedures can be combined with the previously mentioned botulinum toxin procedures for a synergistic effect.
Deoxycholic Acid—Deoxycholic acid is an injectable product used to selectively remove unwanted fat. It currently is approved by the US Food and Drug Administration for submental fat, but some providers are experimenting with off-label uses. Buccal fat pad removal—or in this case reduction by dissolution—tends to give a thinner, more feminine facial appearance.12 Reducing fat around the axillae also can help promote a more masculine upper torso.13 The safety of deoxycholic acid in these areas has not been adequately tested; thus, caution should be used when discussing these off-label uses with patients.
Hair and Tattoo Removal—Hair removal may be desired by TGD persons for a variety of reasons. Because cisgender females tend to have less body hair overall, transgender people in pursuit of a more feminine appearance often desire removal of facial, neck, and body hair. Although shaving and other modalities such as waxing and chemical depilatories are readily available at-home options, they are not permanent and may lead to folliculitis or pseudofolliculitis barbae. Laser hair removal (LHR) and electrolysis are modalities provided by dermatologists that tend to be more permanent and lead to better outcomes, including less irritation and better aesthetic appearance. It is important to keep in mind that not every person and not every body site can be safely treated with LHR. Patients with lighter skin types and darker hair tend to have the most effective response with a higher margin of safety, as these features allow the laser energy to be selectively absorbed by the melanin in the hair bulb and not by the background skin pigmentation.14,15 Inappropriate patient selection or improper settings for wavelength, pulse width, or fluences can lead to burns and permanent scarring.14,15 Electrolysis is an alternative to hair removal within tattoos and is more effective for those individuals with blonde, red, or white hair.16
Another novel treatment for unwanted hair is eflornithine hydrochloride cream, which works by blocking ornithine decarboxylase, the enzyme that stimulates hair growth. It currently is approved to reduce unwanted hair on the face and adjacent areas under the chin; however the effects of this medication are modest and the medication can be expensive.17
Cosmetic hair and tattoo removal are not currently covered by TRICARE, except in cases of surgical and donor-site preparation for some GAPs. Individuals may desire removal of tattoos at surgery sites to obtain more natural-appearing skin. Currently, GAPs such as vaginoplasty, phalloplasty, and metoidioplasty—often referred to by patients as “bottom surgeries”—include insurance coverage for tattoo removal, LHR, and/or electrolysis.
Management of Hormonal Adverse Effects
Acne—Individuals on testosterone supplementation tend to develop acne for the first several years of treatment, but it may improve with time.18 Acne is treated in individuals receiving testosterone the same way as it is treated in cisgender men, with numerous options for topical and oral medications. In trans masculine persons, spironolactone therapy typically is avoided because it may interfere with the actions of exogenous testosterone administered as part of gender-affirming medical treatment and may lead to other undesired adverse effects such as impotence and gynecomastia.1
Although acne typically improves after starting estrogen therapy, patients receiving estrogens may still develop acne. Most trans feminine patients will already be on an estrogen and an antiandrogen, often spironolactone.1 Spironolactone often is used as monotherapy for acne control in cisgender women. Additionally, an important factor to consider with spironolactone is the possible adverse effect of increased micturition. Currently, the military rarely has gender-inclusive restroom options, which can create a challenge for TSMs who find themselves needing to use the restroom more frequently in the workplace.
If planning therapy with isotretinoin, dermatologists should discuss several important factors with all patients, including TGD patients. One consideration is the patient’s planned future surgeries. Although new literature shows that isotretinoin does not adversely affect wound healing,19 some surgeons still adhere to an isotretinoin washout period of 6 to 12 months prior to performing any elective procedures due to concerns about wound healing.20,21 Second, be sure to properly assess and document pregnancy potential in TGD persons. Providers should not assume that a patient is not pregnant or is not trying to become pregnant just because they are trans masculine. It also is important to note that testosterone is not a reliable birth control method.1 If a patient still has ovaries, fallopian tubes, and a uterus, they are considered medically capable of pregnancy, and providers should keep this in mind regarding all procedures in the TGD population.
Another newer acne treatment modality is the 1762-nm laser, which targets sebaceous glands.22 This device allows for targeted treatment of acne-prone areas without systemic therapy such as retinoids or antiandrogens. The 1762-nm laser is not widely available but may become a regular treatment option once its benefits are proven over time.
Alopecia and Hyperpigmentation—Androgens, whether endogenously or exogenously derived, can lead to androgenetic alopecia (AGA) in genetically susceptible individuals. Trans masculine persons and others receiving androgen therapy are at higher risk for AGA, which often is undesirable and may be considered gender affirming by some TGD persons. Standard AGA treatments for cisgender men also can be used in trans masculine persons. Some of the most common anti-AGA medications are topical minoxidil, oral finasteride, and oral minoxidil. Although Coleman et al1 recently reported that finasteride may be an appropriate treatment option in trans masculine persons experiencing alopecia, treatment with 5α-reductase inhibitors may impair clitoral growth and the development of facial and body hair. Further studies are needed to assess the efficacy and safety of 5α-reductase inhibitors in transgender populations.1 Dutasteride may be used off-label and comes with a similar potential adverse-event profile as finasteride, which includes depression, decreased libido, erectile dysfunction, ejaculation disorders, and gynecomastia.
Conversely, AGA tends to improve in trans feminine persons and others receiving estrogen and antiandrogen therapy. Natural testosterone production is suppressed by estrogens and spironolactone as well as in patients who undergo orchiectomy.1 Although spironolactone is not approved for acne, AGA, or hirsutism, it is a standard treatment of AGA in cisgender women because it functions to block the effects of androgens, including at the hair follicle. Finasteride may be used for AGA in cisgender women but it is not recommended for trans feminine persons.1
There are many other modalities available for the treatment of AGA that are less commonly used—some may be cost prohibitive or do not have robust supporting evidence, or both. One example is hair
Melasma is a hyperpigmentation disorder related to estrogens, UV light exposure, and sometimes medication use (eg, hormonal birth control, spironolactone).24 The mainstay of treatment is prevention, including sun avoidance as well as use of sun-protective clothing and broad-spectrum sunscreens. Dermatologists tend to recommend physical sunscreens containing zinc oxide, titanium dioxide, and/or iron oxide, as they cover a wider UV spectrum and also provide some protection from visible light. Once melasma is present, dermatologists still have several treatment options. Topical hydroquinone is a proven treatment; however, it must be used with caution to avoid ochronosis. With careful patient selection, chemical peels also are effective treatment options for dyspigmentation and hyperpigmentation. Energy devices such as intense pulsed light and tattoo removal lasers—Q-switched lasers and picosecond pulse widths—also can be used to treat hyperpigmentation. Oral, intralesional, and topical tranexamic acid are newer treatment options for melasma that still are being studied and have shown promising results. Further studies are needed to determine long-term safety and optimal treatment regimens.24,25
Many insurance carriers, including TRICARE, do not routinely cover medical management of AGA or melasma. Patients should be advised that they likely will have to pay for any medications prescribed and procedures undertaken for these purposes; however, some medication costs can be offset by ordering larger prescription quantities, such as a 90-day supply vs a 30-day supply, as well as utilizing pharmacy discount programs.
Scar Management Following Surgery
In TSMs who undergo gender-affirming surgeries, dermatologists play an important role when scar symptoms develop, including pruritus, tenderness, and/or paresthesia. In the military, some common treatment modalities for symptomatic scars include intralesional steroids with or without 5-fluouroruacil and the fractionated CO2 laser. There also are numerous experimental treatment options for scars, including intralesional or perilesional botulinum toxin, the pulsed dye laser, or nonablative fractionated lasers. These modalities also may be used on hypertrophic scars or keloids. Another option for keloids is scar excision followed by superficial radiation therapy.26
Mental Health Considerations
Providers must take psychological adverse effects into consideration when considering medical therapies for dermatologic conditions in TGD patients. In particular, it is important to consider the risks for increased rates of depression and suicidal ideation formerly associated with the use of isotretinoin and finasteride, though much of the evidence regarding these risks has been called into question in recent years.27,28 Nonetheless, it remains prominent in lay media and may be a more important consideration in patients at higher baseline risk.27 Although there are no known studies that have expressly assessed rates of depression or suicidal ideation in TGD patients taking isotretinoin or finasteride, it is well established that TGD persons are at higher baseline risk for depression and suicidality.1,7,8 All patients should be carefully assessed for depression and suicidal ideation as well as counseled regarding these risks prior to initiating these therapies. If concerns for untreated mental health issues arise during screening and counseling, patients should be referred for assessment by a behavioral health specialist prior to starting therapy.
Future Directions
The future of TGD health care in the military could see an expansion of covered benefits and the development of new dermatologic procedures or medications. Research and policy evolution are necessary to bridge the current gaps in care; however, it is unlikely that all procedures currently considered to be cosmetic will become covered benefits.
Facial LHR is a promising candidate for future coverage for trans feminine persons. When cisgender men develop adverse effects from mandatory daily shaving, LHR is already a covered benefit. Two arguments in support of adding LHR for TGD patients revolve around achieving and maintaining an appearance congruent with their gender along with avoiding unwanted adverse effects related to daily shaving. Visual conformity with one’s affirmed gender has been associated with improvements in well-being, quality of life, and some mental health conditions.29
Scar prevention, treatment, and reduction are additional areas under active research in which dermatologists likely will play a crucial role.30,31 As more dermatologic procedures are performed on TGD persons, the published data and collective knowledge regarding best practices in this population will continue to grow, which will lead to improved cosmetic and safety outcomes.
Final Thoughts
Although dermatologists do not directly perform gender-affirming surgeries or hormone management, they do play an important role in enhancing a TGD person’s desired appearance and managing possible adverse effects resulting from gender-affirming interventions. There have been considerable advancements in TGD health care over the past decade, but there likely are more changes on the way. As policies and understanding of TGD health care needs evolve, it is crucial that the military health care system adapts to provide comprehensive, accessible, and equitable care, which includes expanding the range of covered dermatologic treatments to fully support the health and readiness of TSMs.
Acknowledgment—We would like to extend our sincere appreciation to the invaluable contributions and editorial support provided by Allison Higgins, JD (San Antonio, Texas), throughout the writing of this article.
People whose gender identity differs from the sex assigned at birth are referred to as transgender. For some, gender identity may not fit into the binary constructs of male and female but rather falls between, within, or outside this construct. These people often consider themselves nonbinary or gender diverse. As the terminology continues to evolve, current recommendations include referring to this patient population as transgender and gender diverse (TGD) to ensure the broadest inclusivity.1 In this article, the following terms are used as defined below:
- The terms transgender woman and trans feminine describe persons who were assigned male gender at birth but their affirmed gender is female or nonmasculine.
- The terms transgender man and trans masculine describe persons who were assigned female gender at birth but their affirmed gender is male or nonfeminine.
The US Military’s policies on the service of TGD persons have evolved considerably over the past decade. Initial military policies barred TGD service members (TSMs) from service all together, leading to challenges in accessing necessary health care. The first official memorandum explicitly allowing military service by TGD persons was released on June 30, 2016.2 The intention of this memorandum was 2-fold: (1) to allow TGD persons to serve in the military so long as they meet “the rigorous standards for military service and readiness” by fulfilling the same standards and procedures as other military service members, including medical fitness for duty, physical fitness, uniform and grooming, deployability, and retention, and (2) to direct the establishment of new or updated policies to specific departments and prescribe procedures for retention standards, separation from service, in-service transition, and medical coverage.2 Several other official policies were released following this initial memorandum that provided more specific guidance on how to implement these policies at the level of the force, unit, and individual service member.
Modifications to the original 2016 policies had varying impacts on transgender health care provision and access.3 At the time of publication of this article, the current policy—the Department of Defense Instruction 1300.284—among others, establishes standards and procedures for the process by which active and reserve TSMs may medically, socially, and legally transition genders within the military. The current policy applies to all military branches and serves as the framework by which each branch currently organizes their gender-affirmation processes (GAP).4
There currently are several different GAP models among the military branches.5 Each branch has a different model or approach to implementing the current policy, with varying service-specific processes in place for TSMs to access gender-affirming care; however, this may be changing. The Defense Health Agency is in the process of consolidating and streamlining the GAP across the Department of Defense branches in an effort to optimize costs and ensure uniformity of care. Per the Defense Health Agency Procedural Instruction Number 6025.21 published in May 2023, the proposed consolidated model likely will entail a single central transgender health center that provides oversight and guidance for several regional joint-service gender-affirming medical hubs. Patients would either be managed at the level of the hub or be referred to the central site.5
Herein, we discuss the importance of gender-affirming care and how military and civilian dermatologists can contribute. We also review disparities in health care and identify areas of improvement.
Benefits of Gender-Affirming Care
Gender-affirming procedures are critical for aligning physical appearance with gender identity. Physical appearance is essential for psychological well-being, operational readiness, and the safety of TSMs.6 It is well documented that TGD persons experience suicidal ideation, depression, stigma, discrimination and violence at higher rates than their cisgender peers.7,8 It is important to recognize that transgender identity is not a mental illness, and these elevated rates have been linked to complex trauma, societal stigma, violence, and discrimination.1 Other studies have suggested that increased access to gender-affirming interventions may ameliorate these mental health concerns.1,7-9
The major components of gender-affirming care include hormone therapy, gender confirmation surgery, and mental health care, if needed. These are covered by TRICARE, the health care program for military service members; however, at the time of publication, many of the dermatologic gender-affirming procedures are not covered by TRICARE because they are considered “cosmetic procedures,” which is a term used by insurance companies but does not accurately indicate whether a procedure is medically necessary or not. Newer literature has demonstrated that gender-affirming care positively affects the lives of TGD patients, strengthening the argument that gender-affirming care is a medical necessity and not just cosmetic.1
Aesthetic Procedures in Gender-Affirming Care
Surgeons, including those within the specialties of oto-laryngology, oral and maxillofacial surgery, urology, gynecology, and plastic surgery, provide major gender-affirming interventions; however, dermatologists may offer less invasive solutions that can serve as a temporary experience prior to undergoing more permanent procedures.Hormonally driven disorders including acne, hair loss, and melasma also are managed by dermatologists, along with scar treatment following surgeries.
Because human variation is expansive and subjective, what is considered feminine or masculine may vary by person, group, culture, and country; therefore, it is imperative to ask patients about their individual aesthetic goals and tailor their treatment accordingly. Feminine and masculine are terms that will be used to describe prototypical appearances and are not meant to define a patient’s current state or ultimate goals. The following procedures and medical interventions are where dermatologists can play an important role in TGD persons’ GAPs.
Botulinum Toxin Injections—Botulinum toxin injection is the most common nonsurgical aesthetic procedure performed around the world.10 The selective paralysis afforded by botulinum toxin has several uses for people undergoing transition. Aesthetically, the feminine eyebrow tends to be positioned above the orbital rim and is arched with its apex between the lateral limbus and lateral canthus,11 while the masculine eyebrow tends to be flatter and fuller and runs over the orbital rim without a peak. For people seeking a more feminine appearance, an eyebrow lift with botulinum toxin can help reshape the typical flatter masculine eyebrow to give it lateral lift that often is considered more feminine. The targeted muscle is the superolateral orbicularis oculi, which serves as a depressor on the eyebrow. This can be combined with purposefully avoiding total lateral frontalis paralysis, which leads to a “Spock” brow for extra lift. Conversely, a naturally arched and higher eyebrow can be flattened and lowered by selectively targeting areas of the frontalis muscle.
Broad square jawlines typically are considered a masculine feature and are another area where botulinum toxin can be used to feminize a patient’s facial features. Targeting the masseter muscle induces muscle weakness, which ultimately may result in atrophy after one or more treatment sessions. This atrophy may lead to narrowing of the lower face and thus may lead to a fuller-appearing midface or overall more heart-shaped face. Every individual’s aesthetic goals are unique and therefore should be discussed prior to any treatment.
Dermal Fillers—Dermal fillers are gel-like substances injected under the skin for subtle contouring of the face. Fillers also can be used to help promote a more masculine or feminine appearance. Filler can be placed in the lips to create a fuller, more projected, feminine-appearing lip. Malar cheek and central lower chin filler can be used to help define a heart-shaped face by accentuating the upper portion of the face and creating a more pointed chin, respectively. Alternatively, filler can be used to masculinize the chin by placing it where it can increase jawline squareness and increase anterior jaw projection. Additionally, filler at the angle of the jaw can help accentuate a square facial shape and a more defined jawline. Although not as widely practiced, lateral brow filler can create a heavier-appearing and broader forehead for a more masculine appearance. These procedures can be combined with the previously mentioned botulinum toxin procedures for a synergistic effect.
Deoxycholic Acid—Deoxycholic acid is an injectable product used to selectively remove unwanted fat. It currently is approved by the US Food and Drug Administration for submental fat, but some providers are experimenting with off-label uses. Buccal fat pad removal—or in this case reduction by dissolution—tends to give a thinner, more feminine facial appearance.12 Reducing fat around the axillae also can help promote a more masculine upper torso.13 The safety of deoxycholic acid in these areas has not been adequately tested; thus, caution should be used when discussing these off-label uses with patients.
Hair and Tattoo Removal—Hair removal may be desired by TGD persons for a variety of reasons. Because cisgender females tend to have less body hair overall, transgender people in pursuit of a more feminine appearance often desire removal of facial, neck, and body hair. Although shaving and other modalities such as waxing and chemical depilatories are readily available at-home options, they are not permanent and may lead to folliculitis or pseudofolliculitis barbae. Laser hair removal (LHR) and electrolysis are modalities provided by dermatologists that tend to be more permanent and lead to better outcomes, including less irritation and better aesthetic appearance. It is important to keep in mind that not every person and not every body site can be safely treated with LHR. Patients with lighter skin types and darker hair tend to have the most effective response with a higher margin of safety, as these features allow the laser energy to be selectively absorbed by the melanin in the hair bulb and not by the background skin pigmentation.14,15 Inappropriate patient selection or improper settings for wavelength, pulse width, or fluences can lead to burns and permanent scarring.14,15 Electrolysis is an alternative to hair removal within tattoos and is more effective for those individuals with blonde, red, or white hair.16
Another novel treatment for unwanted hair is eflornithine hydrochloride cream, which works by blocking ornithine decarboxylase, the enzyme that stimulates hair growth. It currently is approved to reduce unwanted hair on the face and adjacent areas under the chin; however the effects of this medication are modest and the medication can be expensive.17
Cosmetic hair and tattoo removal are not currently covered by TRICARE, except in cases of surgical and donor-site preparation for some GAPs. Individuals may desire removal of tattoos at surgery sites to obtain more natural-appearing skin. Currently, GAPs such as vaginoplasty, phalloplasty, and metoidioplasty—often referred to by patients as “bottom surgeries”—include insurance coverage for tattoo removal, LHR, and/or electrolysis.
Management of Hormonal Adverse Effects
Acne—Individuals on testosterone supplementation tend to develop acne for the first several years of treatment, but it may improve with time.18 Acne is treated in individuals receiving testosterone the same way as it is treated in cisgender men, with numerous options for topical and oral medications. In trans masculine persons, spironolactone therapy typically is avoided because it may interfere with the actions of exogenous testosterone administered as part of gender-affirming medical treatment and may lead to other undesired adverse effects such as impotence and gynecomastia.1
Although acne typically improves after starting estrogen therapy, patients receiving estrogens may still develop acne. Most trans feminine patients will already be on an estrogen and an antiandrogen, often spironolactone.1 Spironolactone often is used as monotherapy for acne control in cisgender women. Additionally, an important factor to consider with spironolactone is the possible adverse effect of increased micturition. Currently, the military rarely has gender-inclusive restroom options, which can create a challenge for TSMs who find themselves needing to use the restroom more frequently in the workplace.
If planning therapy with isotretinoin, dermatologists should discuss several important factors with all patients, including TGD patients. One consideration is the patient’s planned future surgeries. Although new literature shows that isotretinoin does not adversely affect wound healing,19 some surgeons still adhere to an isotretinoin washout period of 6 to 12 months prior to performing any elective procedures due to concerns about wound healing.20,21 Second, be sure to properly assess and document pregnancy potential in TGD persons. Providers should not assume that a patient is not pregnant or is not trying to become pregnant just because they are trans masculine. It also is important to note that testosterone is not a reliable birth control method.1 If a patient still has ovaries, fallopian tubes, and a uterus, they are considered medically capable of pregnancy, and providers should keep this in mind regarding all procedures in the TGD population.
Another newer acne treatment modality is the 1762-nm laser, which targets sebaceous glands.22 This device allows for targeted treatment of acne-prone areas without systemic therapy such as retinoids or antiandrogens. The 1762-nm laser is not widely available but may become a regular treatment option once its benefits are proven over time.
Alopecia and Hyperpigmentation—Androgens, whether endogenously or exogenously derived, can lead to androgenetic alopecia (AGA) in genetically susceptible individuals. Trans masculine persons and others receiving androgen therapy are at higher risk for AGA, which often is undesirable and may be considered gender affirming by some TGD persons. Standard AGA treatments for cisgender men also can be used in trans masculine persons. Some of the most common anti-AGA medications are topical minoxidil, oral finasteride, and oral minoxidil. Although Coleman et al1 recently reported that finasteride may be an appropriate treatment option in trans masculine persons experiencing alopecia, treatment with 5α-reductase inhibitors may impair clitoral growth and the development of facial and body hair. Further studies are needed to assess the efficacy and safety of 5α-reductase inhibitors in transgender populations.1 Dutasteride may be used off-label and comes with a similar potential adverse-event profile as finasteride, which includes depression, decreased libido, erectile dysfunction, ejaculation disorders, and gynecomastia.
Conversely, AGA tends to improve in trans feminine persons and others receiving estrogen and antiandrogen therapy. Natural testosterone production is suppressed by estrogens and spironolactone as well as in patients who undergo orchiectomy.1 Although spironolactone is not approved for acne, AGA, or hirsutism, it is a standard treatment of AGA in cisgender women because it functions to block the effects of androgens, including at the hair follicle. Finasteride may be used for AGA in cisgender women but it is not recommended for trans feminine persons.1
There are many other modalities available for the treatment of AGA that are less commonly used—some may be cost prohibitive or do not have robust supporting evidence, or both. One example is hair
Melasma is a hyperpigmentation disorder related to estrogens, UV light exposure, and sometimes medication use (eg, hormonal birth control, spironolactone).24 The mainstay of treatment is prevention, including sun avoidance as well as use of sun-protective clothing and broad-spectrum sunscreens. Dermatologists tend to recommend physical sunscreens containing zinc oxide, titanium dioxide, and/or iron oxide, as they cover a wider UV spectrum and also provide some protection from visible light. Once melasma is present, dermatologists still have several treatment options. Topical hydroquinone is a proven treatment; however, it must be used with caution to avoid ochronosis. With careful patient selection, chemical peels also are effective treatment options for dyspigmentation and hyperpigmentation. Energy devices such as intense pulsed light and tattoo removal lasers—Q-switched lasers and picosecond pulse widths—also can be used to treat hyperpigmentation. Oral, intralesional, and topical tranexamic acid are newer treatment options for melasma that still are being studied and have shown promising results. Further studies are needed to determine long-term safety and optimal treatment regimens.24,25
Many insurance carriers, including TRICARE, do not routinely cover medical management of AGA or melasma. Patients should be advised that they likely will have to pay for any medications prescribed and procedures undertaken for these purposes; however, some medication costs can be offset by ordering larger prescription quantities, such as a 90-day supply vs a 30-day supply, as well as utilizing pharmacy discount programs.
Scar Management Following Surgery
In TSMs who undergo gender-affirming surgeries, dermatologists play an important role when scar symptoms develop, including pruritus, tenderness, and/or paresthesia. In the military, some common treatment modalities for symptomatic scars include intralesional steroids with or without 5-fluouroruacil and the fractionated CO2 laser. There also are numerous experimental treatment options for scars, including intralesional or perilesional botulinum toxin, the pulsed dye laser, or nonablative fractionated lasers. These modalities also may be used on hypertrophic scars or keloids. Another option for keloids is scar excision followed by superficial radiation therapy.26
Mental Health Considerations
Providers must take psychological adverse effects into consideration when considering medical therapies for dermatologic conditions in TGD patients. In particular, it is important to consider the risks for increased rates of depression and suicidal ideation formerly associated with the use of isotretinoin and finasteride, though much of the evidence regarding these risks has been called into question in recent years.27,28 Nonetheless, it remains prominent in lay media and may be a more important consideration in patients at higher baseline risk.27 Although there are no known studies that have expressly assessed rates of depression or suicidal ideation in TGD patients taking isotretinoin or finasteride, it is well established that TGD persons are at higher baseline risk for depression and suicidality.1,7,8 All patients should be carefully assessed for depression and suicidal ideation as well as counseled regarding these risks prior to initiating these therapies. If concerns for untreated mental health issues arise during screening and counseling, patients should be referred for assessment by a behavioral health specialist prior to starting therapy.
Future Directions
The future of TGD health care in the military could see an expansion of covered benefits and the development of new dermatologic procedures or medications. Research and policy evolution are necessary to bridge the current gaps in care; however, it is unlikely that all procedures currently considered to be cosmetic will become covered benefits.
Facial LHR is a promising candidate for future coverage for trans feminine persons. When cisgender men develop adverse effects from mandatory daily shaving, LHR is already a covered benefit. Two arguments in support of adding LHR for TGD patients revolve around achieving and maintaining an appearance congruent with their gender along with avoiding unwanted adverse effects related to daily shaving. Visual conformity with one’s affirmed gender has been associated with improvements in well-being, quality of life, and some mental health conditions.29
Scar prevention, treatment, and reduction are additional areas under active research in which dermatologists likely will play a crucial role.30,31 As more dermatologic procedures are performed on TGD persons, the published data and collective knowledge regarding best practices in this population will continue to grow, which will lead to improved cosmetic and safety outcomes.
Final Thoughts
Although dermatologists do not directly perform gender-affirming surgeries or hormone management, they do play an important role in enhancing a TGD person’s desired appearance and managing possible adverse effects resulting from gender-affirming interventions. There have been considerable advancements in TGD health care over the past decade, but there likely are more changes on the way. As policies and understanding of TGD health care needs evolve, it is crucial that the military health care system adapts to provide comprehensive, accessible, and equitable care, which includes expanding the range of covered dermatologic treatments to fully support the health and readiness of TSMs.
Acknowledgment—We would like to extend our sincere appreciation to the invaluable contributions and editorial support provided by Allison Higgins, JD (San Antonio, Texas), throughout the writing of this article.
- Coleman E, Radix AE, Bouman WP, et al. Standards of care for the health of transgender and gender diverse people, version 8. Int J Transgend Health. 2022;23(suppl 1):S1-S260. doi:10.1080/26895269.2022.2100644
- Secretary of Defense. DTM 16-005—military service of transgender service members. June 30, 2016. Accessed June 17, 2024. https://dod.defense.gov/Portals/1/features/2016/0616_policy/DTM-16-005.pdf
- Office of the Deputy Secretary of Defense. DTM 19-004—military service by transgender persons and persons with gender dysphoria. March 17, 2020. Accessed June 17, 2024. https://health.mil/Reference-Center/Policies/2020/03/17/Military-Service-by-Transgender-Persons-and-Persons-with-Gender-Dysphoria
- Office of the Under Secretary of Defense for Personnel and Readiness. Department of Defense Instruction (DODI) 1300.28. in-service transition for transgender service members. September 4, 2020. Accessed June 17, 2024. https://health.mil/Reference-Center/Policies/2020/09/04/Military-Service-by-Transgender-Persons-and-Persons-with-Gender-Dysphoria
- Defense Health Agency Procedural Instruction Number 6025.21, Guidance for Gender-Affirming Health Care of Transgender and Gender-Diverse Active and Reserve Component Service Members, May 12, 2023. https://www.health.mil/Reference-Center/DHA-Publications/2023/05/12/DHA-PI-6015-21
- Elders MJ, Brown GR, Coleman E, et al. Medical aspects of transgender military service. Armed Forces Soc. 2015;41:199-220. doi:10.1177/0095327X14545625.
- Almazan AN, Keuroghlian AS. Association between gender-affirming surgeries and mental health outcomes. JAMA Surg. 2021;156:611-618.
- Tordoff DM, Wanta JW, Collin A, et al. Mental health outcomes in transgender and nonbinary youths receiving gender-affirming care. JAMA Netw Open. 2022;5:E220978. doi:10.1001/jamanetworkopen.2022.0978
- Olson-Kennedy J, Warus J, Okonta V, et al. Chest reconstruction and chest dysphoria in transmasculine minors and young adults: comparisons of nonsurgical and postsurgical cohorts. JAMA Pediatr. 2018;172:431-436. doi:10.1001/jamapediatrics.2017.5440
- Top non-invasive cosmetic procedures worldwide 2022. Statista website. February 8, 2024. Accessed June 13, 2024. https://www.statista.com/statistics/293449/leading-nonsurgical-cosmetic-procedures/
- Kashkouli MB, Abdolalizadeh P, Abolfathzadeh N, et al. Periorbital facial rejuvenation; applied anatomy and pre-operative assessment. J Curr Ophthalmol. 2017;29:154-168. doi:10.1016/j.joco.2017.04.001
- Thomas MK, D’Silva JA, Borole AJ. Injection lipolysis: a systematic review of literature and our experience with a combination of phosphatidylcholine and deoxycholate over a period of 14 years in 1269 patients of Indian and South East Asian origin. J Cutan Aesthet Surg. 2018;11:222-228. doi:10.4103/JCAS.JCAS_117_18
- Jegasothy SM. Deoxycholic acid injections for bra-line lipolysis. Dermatol Surg. 2018;44:757-760. doi:10.1097/DSS.0000000000001311
- Dierickx CC. Hair removal by lasers and intense pulsed light sources. Dermatol Clin. 2002;20:135-146. doi:10.1016/s0733-8635(03)00052-4
- Lepselter J, Elman M. Biological and clinical aspects in laser hair removal. J Dermatolog Treat. 2004;15:72-83. doi:10.1080/09546630310023152
- Yuan N, Feldman AT, Chin P, et al. Comparison of permanent hair removal procedures before gender-affirming vaginoplasty: why we should consider laser hair removal as a first-line treatment for patients who meet criteria. Sex Med. 2022;10:100545. doi:10.1016/j.esxm.2022.100545
- Kumar A, Naguib YW, Shi YC, et al. A method to improve the efficacy of topical eflornithine hydrochloride cream. Drug Deliv. 2016;23:1495-1501. doi:10.3109/10717544.2014.951746
- Hembree WC, Cohen-Kettenis PT, Gooren L, et al. Endocrine treatment of gender-dysphoric/gender-incongruent persons: an endocrine society clinical practice guideline. J Clin Endocrinol Metabol. 2017;102:3869-3903.
- Hatami P, Balighi K, Asl HN, et al. Isotretinoin and timing of procedural interventions: clinical implications and practical points. J Cosmet Dermatol. 2023;22:2146-2149. doi:10.1111/jocd.15874
- Rubenstein R, Roenigk HH Jr, Stegman SJ, et al. Atypical keloids after dermabrasion of patients taking isotretinoin. J Am Acad Dermatol. 1986;15(2 pt 1):280-285.
- Zachariae H. Delayed wound healing and keloid formation following argon laser treatment or dermabrasion during isotretinoin treatment. Br J Dermatol. 1988;118:703-706.
- Goldberg D, Kothare A, Doucette M, et al. Selective photothermolysis with a novel 1726 nm laser beam: a safe and effective solution for acne vulgaris. J Cosmet Dermatol. 2023;22:486-496. doi:10.1111/jocd.15602
- Sun HY, Sebaratnam DF. Clascoterone as a novel treatment for androgenetic alopecia. Clin Exp Dermatol. 2020;45:913-914. doi:10.1111/ced.14292
- Bolognia JL, Schaffer JV, Cerroni L. Dermatology: 2-Volume Set. Elsevier; 2024:1130.
- Konisky H, Balazic E, Jaller JA, et al. Tranexamic acid in melasma: a focused review on drug administration routes. J Cosmet Dermatol. 2023;22:1197-1206. doi:10.1111/jocd.15589
- Walsh LA, Wu E, Pontes D, et al. Keloid treatments: an evidence-based systematic review of recent advances. Syst Rev. 2023;12:42. doi:10.1186/s13643-023-02192-7
- Kridin K, Ludwig RJ. Isotretinoin and the risk of psychiatric disturbances: a global study shedding new light on a debatable story. J Am Acad Dermatol. 2023;88:388-394. doi:10.1016/j.jaad.2022.10.031
- Dyson TE, Cantrell MA, Lund BC. Lack of association between 5α-reductase inhibitors and depression. J Urol. 2020;204:793-798. doi:10.1097/JU.0000000000001079
- To M, Zhang Q, Bradlyn A, et al. Visual conformity with affirmed gender or “passing”: its distribution and association with depression and anxiety in a cohort of transgender people. J Sex Med. 2020;17:2084-2092. doi:10.1016/j.jsxm.2020.07.019
- Fernandes MG, da Silva LP, Cerqueira MT, et al. Mechanomodulatory biomaterials prospects in scar prevention and treatment. Acta Biomater. 2022;150:22-33. doi:10.1016/j.actbio.2022.07.042
- Kolli H, Moy RL. Prevention of scarring with intraoperative erbium:YAG laser treatment. J Drugs Dermatol. 2020;19:1040-1043. doi:10.36849/JDD.2020.5244
- Coleman E, Radix AE, Bouman WP, et al. Standards of care for the health of transgender and gender diverse people, version 8. Int J Transgend Health. 2022;23(suppl 1):S1-S260. doi:10.1080/26895269.2022.2100644
- Secretary of Defense. DTM 16-005—military service of transgender service members. June 30, 2016. Accessed June 17, 2024. https://dod.defense.gov/Portals/1/features/2016/0616_policy/DTM-16-005.pdf
- Office of the Deputy Secretary of Defense. DTM 19-004—military service by transgender persons and persons with gender dysphoria. March 17, 2020. Accessed June 17, 2024. https://health.mil/Reference-Center/Policies/2020/03/17/Military-Service-by-Transgender-Persons-and-Persons-with-Gender-Dysphoria
- Office of the Under Secretary of Defense for Personnel and Readiness. Department of Defense Instruction (DODI) 1300.28. in-service transition for transgender service members. September 4, 2020. Accessed June 17, 2024. https://health.mil/Reference-Center/Policies/2020/09/04/Military-Service-by-Transgender-Persons-and-Persons-with-Gender-Dysphoria
- Defense Health Agency Procedural Instruction Number 6025.21, Guidance for Gender-Affirming Health Care of Transgender and Gender-Diverse Active and Reserve Component Service Members, May 12, 2023. https://www.health.mil/Reference-Center/DHA-Publications/2023/05/12/DHA-PI-6015-21
- Elders MJ, Brown GR, Coleman E, et al. Medical aspects of transgender military service. Armed Forces Soc. 2015;41:199-220. doi:10.1177/0095327X14545625.
- Almazan AN, Keuroghlian AS. Association between gender-affirming surgeries and mental health outcomes. JAMA Surg. 2021;156:611-618.
- Tordoff DM, Wanta JW, Collin A, et al. Mental health outcomes in transgender and nonbinary youths receiving gender-affirming care. JAMA Netw Open. 2022;5:E220978. doi:10.1001/jamanetworkopen.2022.0978
- Olson-Kennedy J, Warus J, Okonta V, et al. Chest reconstruction and chest dysphoria in transmasculine minors and young adults: comparisons of nonsurgical and postsurgical cohorts. JAMA Pediatr. 2018;172:431-436. doi:10.1001/jamapediatrics.2017.5440
- Top non-invasive cosmetic procedures worldwide 2022. Statista website. February 8, 2024. Accessed June 13, 2024. https://www.statista.com/statistics/293449/leading-nonsurgical-cosmetic-procedures/
- Kashkouli MB, Abdolalizadeh P, Abolfathzadeh N, et al. Periorbital facial rejuvenation; applied anatomy and pre-operative assessment. J Curr Ophthalmol. 2017;29:154-168. doi:10.1016/j.joco.2017.04.001
- Thomas MK, D’Silva JA, Borole AJ. Injection lipolysis: a systematic review of literature and our experience with a combination of phosphatidylcholine and deoxycholate over a period of 14 years in 1269 patients of Indian and South East Asian origin. J Cutan Aesthet Surg. 2018;11:222-228. doi:10.4103/JCAS.JCAS_117_18
- Jegasothy SM. Deoxycholic acid injections for bra-line lipolysis. Dermatol Surg. 2018;44:757-760. doi:10.1097/DSS.0000000000001311
- Dierickx CC. Hair removal by lasers and intense pulsed light sources. Dermatol Clin. 2002;20:135-146. doi:10.1016/s0733-8635(03)00052-4
- Lepselter J, Elman M. Biological and clinical aspects in laser hair removal. J Dermatolog Treat. 2004;15:72-83. doi:10.1080/09546630310023152
- Yuan N, Feldman AT, Chin P, et al. Comparison of permanent hair removal procedures before gender-affirming vaginoplasty: why we should consider laser hair removal as a first-line treatment for patients who meet criteria. Sex Med. 2022;10:100545. doi:10.1016/j.esxm.2022.100545
- Kumar A, Naguib YW, Shi YC, et al. A method to improve the efficacy of topical eflornithine hydrochloride cream. Drug Deliv. 2016;23:1495-1501. doi:10.3109/10717544.2014.951746
- Hembree WC, Cohen-Kettenis PT, Gooren L, et al. Endocrine treatment of gender-dysphoric/gender-incongruent persons: an endocrine society clinical practice guideline. J Clin Endocrinol Metabol. 2017;102:3869-3903.
- Hatami P, Balighi K, Asl HN, et al. Isotretinoin and timing of procedural interventions: clinical implications and practical points. J Cosmet Dermatol. 2023;22:2146-2149. doi:10.1111/jocd.15874
- Rubenstein R, Roenigk HH Jr, Stegman SJ, et al. Atypical keloids after dermabrasion of patients taking isotretinoin. J Am Acad Dermatol. 1986;15(2 pt 1):280-285.
- Zachariae H. Delayed wound healing and keloid formation following argon laser treatment or dermabrasion during isotretinoin treatment. Br J Dermatol. 1988;118:703-706.
- Goldberg D, Kothare A, Doucette M, et al. Selective photothermolysis with a novel 1726 nm laser beam: a safe and effective solution for acne vulgaris. J Cosmet Dermatol. 2023;22:486-496. doi:10.1111/jocd.15602
- Sun HY, Sebaratnam DF. Clascoterone as a novel treatment for androgenetic alopecia. Clin Exp Dermatol. 2020;45:913-914. doi:10.1111/ced.14292
- Bolognia JL, Schaffer JV, Cerroni L. Dermatology: 2-Volume Set. Elsevier; 2024:1130.
- Konisky H, Balazic E, Jaller JA, et al. Tranexamic acid in melasma: a focused review on drug administration routes. J Cosmet Dermatol. 2023;22:1197-1206. doi:10.1111/jocd.15589
- Walsh LA, Wu E, Pontes D, et al. Keloid treatments: an evidence-based systematic review of recent advances. Syst Rev. 2023;12:42. doi:10.1186/s13643-023-02192-7
- Kridin K, Ludwig RJ. Isotretinoin and the risk of psychiatric disturbances: a global study shedding new light on a debatable story. J Am Acad Dermatol. 2023;88:388-394. doi:10.1016/j.jaad.2022.10.031
- Dyson TE, Cantrell MA, Lund BC. Lack of association between 5α-reductase inhibitors and depression. J Urol. 2020;204:793-798. doi:10.1097/JU.0000000000001079
- To M, Zhang Q, Bradlyn A, et al. Visual conformity with affirmed gender or “passing”: its distribution and association with depression and anxiety in a cohort of transgender people. J Sex Med. 2020;17:2084-2092. doi:10.1016/j.jsxm.2020.07.019
- Fernandes MG, da Silva LP, Cerqueira MT, et al. Mechanomodulatory biomaterials prospects in scar prevention and treatment. Acta Biomater. 2022;150:22-33. doi:10.1016/j.actbio.2022.07.042
- Kolli H, Moy RL. Prevention of scarring with intraoperative erbium:YAG laser treatment. J Drugs Dermatol. 2020;19:1040-1043. doi:10.36849/JDD.2020.5244
Practice Points
- Transgender and gender diverse (TGD) health care is multidisciplinary, and both military and civilian dermatologists can serve an important role.
- Although dermatologists do not directly perform gender-affirming surgeries or hormone management, there are a number of dermatologic procedures and medical interventions that can enhance a TGD person’s desired appearance.
- Dermatologists also can help manage possible adverse effects from gender-affirming interventions.
Benzoyl Peroxide, Benzene, and Lots of Unanswered Questions: Where Are We Now?
March 2024 proved to be a busy month for benzoyl peroxide in the media! We are now at almost 4 months since Valisure, an independent analytical laboratory located in Connecticut, filed a Citizen Petition on benzene in benzoyl peroxide drug products with the US Food and Drug Administration (FDA) on March 5, 2024.1 This petition was filed shortly before the annual meeting of the American Academy of Dermatology was held in San Diego, California, creating quite a stir of concern in the dermatology world. Further information on the degradation of benzoyl peroxide with production of benzene was published in the medical literature in March 2024.2 As benzene is recognized as a human carcinogen, manufacturing regulations exist to assure that it does not appear in topical products either through contamination or degradation over the course of a product’s shelf-life.3
As anticipated, several opinions and commentaries appeared quickly, both on video and in various articles. The American Acne & Rosacea Society (AARS) released a statement on this issue on March 20, 2024.4 The safety of the public is the overarching primary concern. This AARS statement does include some general suggestions related to benzoyl peroxide use based on the best assessment to date while awaiting further guidance from the FDA on this issue. Benzoyl peroxide is approved for use by the FDA as an over-the-counter (OTC) topical product for acne and also is in several FDA-approved prescription topical products.5,6
The following reflects my personal viewpoint as both a dermatologist and a grandfather who has grandchildren who use acne products. My views are not necessarily those of AARS. Since early March 2024, I have read several documents and spoken to several dermatologists, scientists, and formulators with knowledge in this area, including contacts at Valisure. I was hoping to get to some reasonable definitive answer but have not been able to achieve this to my full satisfaction. There are many opinions and concerns, and each one makes sense based on the vantage point of the presenter. However, several unanswered questions remain related to what testing and data are currently required of companies to gain FDA approval of a benzoyl peroxide product, including:
- assessment of stability and degradation products (including benzene),
- validation of testing methods,
- the issue of benzoyl peroxide stability in commercial products, and
- the relevant magnitude of resultant benzene exposures, especially as we are all exposed to benzene from several sources each day.
I am certain that companies with benzoyl peroxide products will evaluate their already-approved products and also do further testing. However, in this situation, which impacts millions of people on so many levels, I feel there needs to be an organized approach to evaluate and resolve the issue, otherwise the likelihood of continued confusion and uncertainty is high. As the FDA is the approval body, I am hoping it will provide definitive guidance within a reasonable timeline so that clinicians, patients, and manufacturers of benzoyl peroxide can proceed with full confidence. Right now, we all remain in a state of limbo. It is time for less talk and more definitive action to sort out this issue.
- Valisure Citizen Petition on Benzene in Benzoyl Peroxide Products. March 5, 2024. Accessed June 5, 2024. https://assets-global.website-files.com/6215052733f8bb8fea016220/65e8560962ed23f744902a7b_Valisure%20Citizen%20Petition%20on%20Benzene%20in%20Benzoyl%20Peroxide%20Drug%20Products.pdf
- Kucera K, Zenzola N, Hudspeth A, et al. Benzoyl peroxide drug products form benzene. Environ Health Perspect. 2024;132:37702. doi:10.1289/EHP13984
- US Food and Drug Administration. Reformulating drug products that contain carbomers manufactured with benzene. December 2023. Accessed June 12, 2024. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/reformulating-drug-products-contain-carbomers-manufactured-benzene
- American Acne & Rosacea Society. Response Statement from the AARS to the Valisure Citizen Petition on Benzene in Benzoyl Peroxide Drug Products. March 20, 2024. Accessed June 12, 2024. https://www.einpresswire.com/article/697481595/response-statement-from-the-aars-to-the-valisure-citizen-petition-on-benzene-in-benzoyl-peroxide-drug-products
- Department of Health and Human Services. Classification of benzoyl peroxide as safe and effective and revision of labeling to drug facts format; topical acne drug products for over-the-counter human use; Final Rule. Fed Registr. 2010;75:9767-9777.
- US Food and Drug Administration. Topical acne drug products for over-the-counter human use—revision of labeling and classification of benzoyl peroxide as safe and effective. June 2011. Accessed June 12, 2024. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/topical-acne-drug-products-over-counter-human-use-revision-labeling-and-classification-benzoyl
March 2024 proved to be a busy month for benzoyl peroxide in the media! We are now at almost 4 months since Valisure, an independent analytical laboratory located in Connecticut, filed a Citizen Petition on benzene in benzoyl peroxide drug products with the US Food and Drug Administration (FDA) on March 5, 2024.1 This petition was filed shortly before the annual meeting of the American Academy of Dermatology was held in San Diego, California, creating quite a stir of concern in the dermatology world. Further information on the degradation of benzoyl peroxide with production of benzene was published in the medical literature in March 2024.2 As benzene is recognized as a human carcinogen, manufacturing regulations exist to assure that it does not appear in topical products either through contamination or degradation over the course of a product’s shelf-life.3
As anticipated, several opinions and commentaries appeared quickly, both on video and in various articles. The American Acne & Rosacea Society (AARS) released a statement on this issue on March 20, 2024.4 The safety of the public is the overarching primary concern. This AARS statement does include some general suggestions related to benzoyl peroxide use based on the best assessment to date while awaiting further guidance from the FDA on this issue. Benzoyl peroxide is approved for use by the FDA as an over-the-counter (OTC) topical product for acne and also is in several FDA-approved prescription topical products.5,6
The following reflects my personal viewpoint as both a dermatologist and a grandfather who has grandchildren who use acne products. My views are not necessarily those of AARS. Since early March 2024, I have read several documents and spoken to several dermatologists, scientists, and formulators with knowledge in this area, including contacts at Valisure. I was hoping to get to some reasonable definitive answer but have not been able to achieve this to my full satisfaction. There are many opinions and concerns, and each one makes sense based on the vantage point of the presenter. However, several unanswered questions remain related to what testing and data are currently required of companies to gain FDA approval of a benzoyl peroxide product, including:
- assessment of stability and degradation products (including benzene),
- validation of testing methods,
- the issue of benzoyl peroxide stability in commercial products, and
- the relevant magnitude of resultant benzene exposures, especially as we are all exposed to benzene from several sources each day.
I am certain that companies with benzoyl peroxide products will evaluate their already-approved products and also do further testing. However, in this situation, which impacts millions of people on so many levels, I feel there needs to be an organized approach to evaluate and resolve the issue, otherwise the likelihood of continued confusion and uncertainty is high. As the FDA is the approval body, I am hoping it will provide definitive guidance within a reasonable timeline so that clinicians, patients, and manufacturers of benzoyl peroxide can proceed with full confidence. Right now, we all remain in a state of limbo. It is time for less talk and more definitive action to sort out this issue.
March 2024 proved to be a busy month for benzoyl peroxide in the media! We are now at almost 4 months since Valisure, an independent analytical laboratory located in Connecticut, filed a Citizen Petition on benzene in benzoyl peroxide drug products with the US Food and Drug Administration (FDA) on March 5, 2024.1 This petition was filed shortly before the annual meeting of the American Academy of Dermatology was held in San Diego, California, creating quite a stir of concern in the dermatology world. Further information on the degradation of benzoyl peroxide with production of benzene was published in the medical literature in March 2024.2 As benzene is recognized as a human carcinogen, manufacturing regulations exist to assure that it does not appear in topical products either through contamination or degradation over the course of a product’s shelf-life.3
As anticipated, several opinions and commentaries appeared quickly, both on video and in various articles. The American Acne & Rosacea Society (AARS) released a statement on this issue on March 20, 2024.4 The safety of the public is the overarching primary concern. This AARS statement does include some general suggestions related to benzoyl peroxide use based on the best assessment to date while awaiting further guidance from the FDA on this issue. Benzoyl peroxide is approved for use by the FDA as an over-the-counter (OTC) topical product for acne and also is in several FDA-approved prescription topical products.5,6
The following reflects my personal viewpoint as both a dermatologist and a grandfather who has grandchildren who use acne products. My views are not necessarily those of AARS. Since early March 2024, I have read several documents and spoken to several dermatologists, scientists, and formulators with knowledge in this area, including contacts at Valisure. I was hoping to get to some reasonable definitive answer but have not been able to achieve this to my full satisfaction. There are many opinions and concerns, and each one makes sense based on the vantage point of the presenter. However, several unanswered questions remain related to what testing and data are currently required of companies to gain FDA approval of a benzoyl peroxide product, including:
- assessment of stability and degradation products (including benzene),
- validation of testing methods,
- the issue of benzoyl peroxide stability in commercial products, and
- the relevant magnitude of resultant benzene exposures, especially as we are all exposed to benzene from several sources each day.
I am certain that companies with benzoyl peroxide products will evaluate their already-approved products and also do further testing. However, in this situation, which impacts millions of people on so many levels, I feel there needs to be an organized approach to evaluate and resolve the issue, otherwise the likelihood of continued confusion and uncertainty is high. As the FDA is the approval body, I am hoping it will provide definitive guidance within a reasonable timeline so that clinicians, patients, and manufacturers of benzoyl peroxide can proceed with full confidence. Right now, we all remain in a state of limbo. It is time for less talk and more definitive action to sort out this issue.
- Valisure Citizen Petition on Benzene in Benzoyl Peroxide Products. March 5, 2024. Accessed June 5, 2024. https://assets-global.website-files.com/6215052733f8bb8fea016220/65e8560962ed23f744902a7b_Valisure%20Citizen%20Petition%20on%20Benzene%20in%20Benzoyl%20Peroxide%20Drug%20Products.pdf
- Kucera K, Zenzola N, Hudspeth A, et al. Benzoyl peroxide drug products form benzene. Environ Health Perspect. 2024;132:37702. doi:10.1289/EHP13984
- US Food and Drug Administration. Reformulating drug products that contain carbomers manufactured with benzene. December 2023. Accessed June 12, 2024. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/reformulating-drug-products-contain-carbomers-manufactured-benzene
- American Acne & Rosacea Society. Response Statement from the AARS to the Valisure Citizen Petition on Benzene in Benzoyl Peroxide Drug Products. March 20, 2024. Accessed June 12, 2024. https://www.einpresswire.com/article/697481595/response-statement-from-the-aars-to-the-valisure-citizen-petition-on-benzene-in-benzoyl-peroxide-drug-products
- Department of Health and Human Services. Classification of benzoyl peroxide as safe and effective and revision of labeling to drug facts format; topical acne drug products for over-the-counter human use; Final Rule. Fed Registr. 2010;75:9767-9777.
- US Food and Drug Administration. Topical acne drug products for over-the-counter human use—revision of labeling and classification of benzoyl peroxide as safe and effective. June 2011. Accessed June 12, 2024. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/topical-acne-drug-products-over-counter-human-use-revision-labeling-and-classification-benzoyl
- Valisure Citizen Petition on Benzene in Benzoyl Peroxide Products. March 5, 2024. Accessed June 5, 2024. https://assets-global.website-files.com/6215052733f8bb8fea016220/65e8560962ed23f744902a7b_Valisure%20Citizen%20Petition%20on%20Benzene%20in%20Benzoyl%20Peroxide%20Drug%20Products.pdf
- Kucera K, Zenzola N, Hudspeth A, et al. Benzoyl peroxide drug products form benzene. Environ Health Perspect. 2024;132:37702. doi:10.1289/EHP13984
- US Food and Drug Administration. Reformulating drug products that contain carbomers manufactured with benzene. December 2023. Accessed June 12, 2024. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/reformulating-drug-products-contain-carbomers-manufactured-benzene
- American Acne & Rosacea Society. Response Statement from the AARS to the Valisure Citizen Petition on Benzene in Benzoyl Peroxide Drug Products. March 20, 2024. Accessed June 12, 2024. https://www.einpresswire.com/article/697481595/response-statement-from-the-aars-to-the-valisure-citizen-petition-on-benzene-in-benzoyl-peroxide-drug-products
- Department of Health and Human Services. Classification of benzoyl peroxide as safe and effective and revision of labeling to drug facts format; topical acne drug products for over-the-counter human use; Final Rule. Fed Registr. 2010;75:9767-9777.
- US Food and Drug Administration. Topical acne drug products for over-the-counter human use—revision of labeling and classification of benzoyl peroxide as safe and effective. June 2011. Accessed June 12, 2024. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/topical-acne-drug-products-over-counter-human-use-revision-labeling-and-classification-benzoyl
Isotretinoin-Induced Skin Fragility in an Aerialist
Isotretinoin was introduced more than 3 decades ago and marked a major advancement in the treatment of severe refractory cystic acne. The most common adverse effects linked to isotretinoin usage are mucocutaneous in nature, manifesting as xerosis and cheilitis.1 Skin fragility and poor wound healing also have been reported.2-6 Current recommendations for avoiding these adverse effects include refraining from waxing, laser procedures, and other elective cutaneous procedures for at least 6 months.7 We present a case of isotretinoin-induced cutaneous fragility resulting in blistering and erosions on the palms of a competitive aerial trapeze artist.
Case Report
A 25-year-old woman presented for follow-up during week 12 of isotretinoin therapy (40 mg twice daily) prescribed for acne. She reported peeling of the skin on the palms following intense aerial acrobatic workouts. She had been a performing aerialist for many years and had never sustained a similar injury. The wounds were painful and led to decreased activity. She had no notable medical history. Physical examination of the palms revealed erosions in a distribution that corresponded to horizontal bar contact and friction (Figure). The patient was advised on proper wound care, application of emollients, and minimizing friction. She completed the course of isotretinoin and has continued aerialist activity without recurrence of skin fragility.
Comment
Skin fragility is a well-known adverse effect of isotretinoin therapy.8 Pavlis and Lieblich9 reported skin fragility in a young wrestler who experienced similar skin erosions due to isotretinoin therapy. The proposed mechanism of isotretinoin-induced skin fragility is multifactorial. It involves an apoptotic effect on sebocytes,5 which results in reduced stratum corneum hydration and an associated increase in transepidermal water loss.6,10,11 Retinoids also are known to cause thinning of the skin, likely due to the disadhesion of both the epidermis and the stratum corneum, which was demonstrated by the easy removal of cornified cells through tape stripping in hairless mice treated with isotretinoin.12 In further investigations, human patients and hairless mice treated with isotretinoin readily developed friction blisters through pencil eraser abrasion.13 Examination of the friction blisters using light and electron microscopy revealed fraying or loss of the stratum corneum and viable epidermis as well as loss of desmosomes and tonofilaments. Additionally, intracellular and intercellular deposits of an unidentified amorphous material were noted.13

Overall, the origin of skin fragility induced by isotretinoin is supported by its effect on sebocytes, increased transepidermal water loss, and profound disruption of the integrity of the epidermis, resulting in an elevated risk for inadvertent skin damage. Patients were encouraged to avoid cosmetic procedures in prior case reports,14-16 and because our case demonstrates the risk for cutaneous injury in athletes due to isotretinoin-induced skin fragility, we propose an extension of these warnings to encompass athletes receiving isotretinoin treatment. Offering early guidance on wound prevention is of paramount importance in maintaining athletic performance and minimizing painful injuries.
- Rajput I, Anjankar VP. Side effects of treating acne vulgaris with isotretinoin: a systematic review. Cureus. 2024;16:E55946. doi:10.7759/cureus.55946
- Hatami P, Balighi K, Asl HN, et al. Isotretinoin and timing of procedural interventions: clinical implications and practical points. J Cosmet Dermatol. 2023;22:2146-2149. doi:10.1111/jocd.15874
- McDonald KA, Shelley AJ, Alavi A. A systematic review on oral isotretinoin therapy and clinically observable wound healing in acne patients. J Cutan Med Surg. 2017;21:325-333. doi:10.1177/1203475417701419
- Layton A. The use of isotretinoin in acne. Dermatoendocrinol. 2009;1:162-169. doi:10.4161/derm.1.3.9364
- Zouboulis CC. Isotretinoin revisited: pluripotent effects on human sebaceous gland cells. J Invest Dermatol. 2006;126:2154-2156. doi:10.1038/sj.jid.5700418
- Kmiec´ ML, Pajor A, Broniarczyk-Dyła G. Evaluation of biophysical skin parameters and assessment of hair growth in patients with acne treated with isotretinoin. Postepy Dermatol Alergol. 2013;30:343-349. doi:10.5114/pdia.2013.39432
- Waldman A, Bolotin D, Arndt KA, et al. ASDS Guidelines Task Force: Consensus recommendations regarding the safety of lasers, dermabrasion, chemical peels, energy devices, and skin surgery during and after isotretinoin use. Dermatolog Surg. 2017;43:1249-1262. doi:10.1097/DSS.0000000000001166
- Aksoy H, Aksoy B, Calikoglu E. Systemic retinoids and scar dehiscence. Indian J Dermatol. 2019;64:68. doi:10.4103/ijd.IJD_148_18
- Pavlis MB, Lieblich L. Isotretinoin-induced skin fragility in a teenaged athlete: a case report. Cutis. 2013;92:33-34.
- Herane MI, Fuenzalida H, Zegpi E, et al. Specific gel-cream as adjuvant to oral isotretinoin improved hydration and prevented TEWL increase—a double-blind, randomized, placebo-controlled study. J Cosmet Dermatol. 2009;8:181-185. doi:10.1111/j.1473-2165.2009.00455.x
- Park KY, Ko EJ, Kim IS, et al. The effect of evening primrose oil for the prevention of xerotic cheilitis in acne patients being treated with isotretinoin: a pilot study. Ann Dermatol. 2014;26:706-712. doi:10.5021/ad.2014.26.6.706
- Elias PM, Fritsch PO, Lampe M, et al. Retinoid effects on epidermal structure, differentiation, and permeability. Lab Invest. 1981;44:531-540.
- Williams ML, Elias PM. Nature of skin fragility in patients receiving retinoids for systemic effect. Arch Dermatol. 1981;117:611-619.
- Rubenstein R, Roenigk HH, Stegman SJ, et al. Atypical keloids after dermabrasion of patients taking isotretinoin. J Am Acad Dermatol. 1986;15:280-285. doi:10.1016/S0190-9622(86)70167-9
- Zachariae H. Delayed wound healing and keloid formation following argon laser treatment or dermabrasion during isotretinoin treatment. Br J Dermatol. 1988;118:703-706. doi:10.1111/j.1365-2133.1988.tb02574.x
- Katz BE, Mac Farlane DF. Atypical facial scarring after isotretinoin therapy in a patient with previous dermabrasion. J Am Acad Dermatol. 1994;30:852-853. doi:10.1016/S0190-9622(94)70096-6
Isotretinoin was introduced more than 3 decades ago and marked a major advancement in the treatment of severe refractory cystic acne. The most common adverse effects linked to isotretinoin usage are mucocutaneous in nature, manifesting as xerosis and cheilitis.1 Skin fragility and poor wound healing also have been reported.2-6 Current recommendations for avoiding these adverse effects include refraining from waxing, laser procedures, and other elective cutaneous procedures for at least 6 months.7 We present a case of isotretinoin-induced cutaneous fragility resulting in blistering and erosions on the palms of a competitive aerial trapeze artist.
Case Report
A 25-year-old woman presented for follow-up during week 12 of isotretinoin therapy (40 mg twice daily) prescribed for acne. She reported peeling of the skin on the palms following intense aerial acrobatic workouts. She had been a performing aerialist for many years and had never sustained a similar injury. The wounds were painful and led to decreased activity. She had no notable medical history. Physical examination of the palms revealed erosions in a distribution that corresponded to horizontal bar contact and friction (Figure). The patient was advised on proper wound care, application of emollients, and minimizing friction. She completed the course of isotretinoin and has continued aerialist activity without recurrence of skin fragility.
Comment
Skin fragility is a well-known adverse effect of isotretinoin therapy.8 Pavlis and Lieblich9 reported skin fragility in a young wrestler who experienced similar skin erosions due to isotretinoin therapy. The proposed mechanism of isotretinoin-induced skin fragility is multifactorial. It involves an apoptotic effect on sebocytes,5 which results in reduced stratum corneum hydration and an associated increase in transepidermal water loss.6,10,11 Retinoids also are known to cause thinning of the skin, likely due to the disadhesion of both the epidermis and the stratum corneum, which was demonstrated by the easy removal of cornified cells through tape stripping in hairless mice treated with isotretinoin.12 In further investigations, human patients and hairless mice treated with isotretinoin readily developed friction blisters through pencil eraser abrasion.13 Examination of the friction blisters using light and electron microscopy revealed fraying or loss of the stratum corneum and viable epidermis as well as loss of desmosomes and tonofilaments. Additionally, intracellular and intercellular deposits of an unidentified amorphous material were noted.13

Overall, the origin of skin fragility induced by isotretinoin is supported by its effect on sebocytes, increased transepidermal water loss, and profound disruption of the integrity of the epidermis, resulting in an elevated risk for inadvertent skin damage. Patients were encouraged to avoid cosmetic procedures in prior case reports,14-16 and because our case demonstrates the risk for cutaneous injury in athletes due to isotretinoin-induced skin fragility, we propose an extension of these warnings to encompass athletes receiving isotretinoin treatment. Offering early guidance on wound prevention is of paramount importance in maintaining athletic performance and minimizing painful injuries.
Isotretinoin was introduced more than 3 decades ago and marked a major advancement in the treatment of severe refractory cystic acne. The most common adverse effects linked to isotretinoin usage are mucocutaneous in nature, manifesting as xerosis and cheilitis.1 Skin fragility and poor wound healing also have been reported.2-6 Current recommendations for avoiding these adverse effects include refraining from waxing, laser procedures, and other elective cutaneous procedures for at least 6 months.7 We present a case of isotretinoin-induced cutaneous fragility resulting in blistering and erosions on the palms of a competitive aerial trapeze artist.
Case Report
A 25-year-old woman presented for follow-up during week 12 of isotretinoin therapy (40 mg twice daily) prescribed for acne. She reported peeling of the skin on the palms following intense aerial acrobatic workouts. She had been a performing aerialist for many years and had never sustained a similar injury. The wounds were painful and led to decreased activity. She had no notable medical history. Physical examination of the palms revealed erosions in a distribution that corresponded to horizontal bar contact and friction (Figure). The patient was advised on proper wound care, application of emollients, and minimizing friction. She completed the course of isotretinoin and has continued aerialist activity without recurrence of skin fragility.
Comment
Skin fragility is a well-known adverse effect of isotretinoin therapy.8 Pavlis and Lieblich9 reported skin fragility in a young wrestler who experienced similar skin erosions due to isotretinoin therapy. The proposed mechanism of isotretinoin-induced skin fragility is multifactorial. It involves an apoptotic effect on sebocytes,5 which results in reduced stratum corneum hydration and an associated increase in transepidermal water loss.6,10,11 Retinoids also are known to cause thinning of the skin, likely due to the disadhesion of both the epidermis and the stratum corneum, which was demonstrated by the easy removal of cornified cells through tape stripping in hairless mice treated with isotretinoin.12 In further investigations, human patients and hairless mice treated with isotretinoin readily developed friction blisters through pencil eraser abrasion.13 Examination of the friction blisters using light and electron microscopy revealed fraying or loss of the stratum corneum and viable epidermis as well as loss of desmosomes and tonofilaments. Additionally, intracellular and intercellular deposits of an unidentified amorphous material were noted.13

Overall, the origin of skin fragility induced by isotretinoin is supported by its effect on sebocytes, increased transepidermal water loss, and profound disruption of the integrity of the epidermis, resulting in an elevated risk for inadvertent skin damage. Patients were encouraged to avoid cosmetic procedures in prior case reports,14-16 and because our case demonstrates the risk for cutaneous injury in athletes due to isotretinoin-induced skin fragility, we propose an extension of these warnings to encompass athletes receiving isotretinoin treatment. Offering early guidance on wound prevention is of paramount importance in maintaining athletic performance and minimizing painful injuries.
- Rajput I, Anjankar VP. Side effects of treating acne vulgaris with isotretinoin: a systematic review. Cureus. 2024;16:E55946. doi:10.7759/cureus.55946
- Hatami P, Balighi K, Asl HN, et al. Isotretinoin and timing of procedural interventions: clinical implications and practical points. J Cosmet Dermatol. 2023;22:2146-2149. doi:10.1111/jocd.15874
- McDonald KA, Shelley AJ, Alavi A. A systematic review on oral isotretinoin therapy and clinically observable wound healing in acne patients. J Cutan Med Surg. 2017;21:325-333. doi:10.1177/1203475417701419
- Layton A. The use of isotretinoin in acne. Dermatoendocrinol. 2009;1:162-169. doi:10.4161/derm.1.3.9364
- Zouboulis CC. Isotretinoin revisited: pluripotent effects on human sebaceous gland cells. J Invest Dermatol. 2006;126:2154-2156. doi:10.1038/sj.jid.5700418
- Kmiec´ ML, Pajor A, Broniarczyk-Dyła G. Evaluation of biophysical skin parameters and assessment of hair growth in patients with acne treated with isotretinoin. Postepy Dermatol Alergol. 2013;30:343-349. doi:10.5114/pdia.2013.39432
- Waldman A, Bolotin D, Arndt KA, et al. ASDS Guidelines Task Force: Consensus recommendations regarding the safety of lasers, dermabrasion, chemical peels, energy devices, and skin surgery during and after isotretinoin use. Dermatolog Surg. 2017;43:1249-1262. doi:10.1097/DSS.0000000000001166
- Aksoy H, Aksoy B, Calikoglu E. Systemic retinoids and scar dehiscence. Indian J Dermatol. 2019;64:68. doi:10.4103/ijd.IJD_148_18
- Pavlis MB, Lieblich L. Isotretinoin-induced skin fragility in a teenaged athlete: a case report. Cutis. 2013;92:33-34.
- Herane MI, Fuenzalida H, Zegpi E, et al. Specific gel-cream as adjuvant to oral isotretinoin improved hydration and prevented TEWL increase—a double-blind, randomized, placebo-controlled study. J Cosmet Dermatol. 2009;8:181-185. doi:10.1111/j.1473-2165.2009.00455.x
- Park KY, Ko EJ, Kim IS, et al. The effect of evening primrose oil for the prevention of xerotic cheilitis in acne patients being treated with isotretinoin: a pilot study. Ann Dermatol. 2014;26:706-712. doi:10.5021/ad.2014.26.6.706
- Elias PM, Fritsch PO, Lampe M, et al. Retinoid effects on epidermal structure, differentiation, and permeability. Lab Invest. 1981;44:531-540.
- Williams ML, Elias PM. Nature of skin fragility in patients receiving retinoids for systemic effect. Arch Dermatol. 1981;117:611-619.
- Rubenstein R, Roenigk HH, Stegman SJ, et al. Atypical keloids after dermabrasion of patients taking isotretinoin. J Am Acad Dermatol. 1986;15:280-285. doi:10.1016/S0190-9622(86)70167-9
- Zachariae H. Delayed wound healing and keloid formation following argon laser treatment or dermabrasion during isotretinoin treatment. Br J Dermatol. 1988;118:703-706. doi:10.1111/j.1365-2133.1988.tb02574.x
- Katz BE, Mac Farlane DF. Atypical facial scarring after isotretinoin therapy in a patient with previous dermabrasion. J Am Acad Dermatol. 1994;30:852-853. doi:10.1016/S0190-9622(94)70096-6
- Rajput I, Anjankar VP. Side effects of treating acne vulgaris with isotretinoin: a systematic review. Cureus. 2024;16:E55946. doi:10.7759/cureus.55946
- Hatami P, Balighi K, Asl HN, et al. Isotretinoin and timing of procedural interventions: clinical implications and practical points. J Cosmet Dermatol. 2023;22:2146-2149. doi:10.1111/jocd.15874
- McDonald KA, Shelley AJ, Alavi A. A systematic review on oral isotretinoin therapy and clinically observable wound healing in acne patients. J Cutan Med Surg. 2017;21:325-333. doi:10.1177/1203475417701419
- Layton A. The use of isotretinoin in acne. Dermatoendocrinol. 2009;1:162-169. doi:10.4161/derm.1.3.9364
- Zouboulis CC. Isotretinoin revisited: pluripotent effects on human sebaceous gland cells. J Invest Dermatol. 2006;126:2154-2156. doi:10.1038/sj.jid.5700418
- Kmiec´ ML, Pajor A, Broniarczyk-Dyła G. Evaluation of biophysical skin parameters and assessment of hair growth in patients with acne treated with isotretinoin. Postepy Dermatol Alergol. 2013;30:343-349. doi:10.5114/pdia.2013.39432
- Waldman A, Bolotin D, Arndt KA, et al. ASDS Guidelines Task Force: Consensus recommendations regarding the safety of lasers, dermabrasion, chemical peels, energy devices, and skin surgery during and after isotretinoin use. Dermatolog Surg. 2017;43:1249-1262. doi:10.1097/DSS.0000000000001166
- Aksoy H, Aksoy B, Calikoglu E. Systemic retinoids and scar dehiscence. Indian J Dermatol. 2019;64:68. doi:10.4103/ijd.IJD_148_18
- Pavlis MB, Lieblich L. Isotretinoin-induced skin fragility in a teenaged athlete: a case report. Cutis. 2013;92:33-34.
- Herane MI, Fuenzalida H, Zegpi E, et al. Specific gel-cream as adjuvant to oral isotretinoin improved hydration and prevented TEWL increase—a double-blind, randomized, placebo-controlled study. J Cosmet Dermatol. 2009;8:181-185. doi:10.1111/j.1473-2165.2009.00455.x
- Park KY, Ko EJ, Kim IS, et al. The effect of evening primrose oil for the prevention of xerotic cheilitis in acne patients being treated with isotretinoin: a pilot study. Ann Dermatol. 2014;26:706-712. doi:10.5021/ad.2014.26.6.706
- Elias PM, Fritsch PO, Lampe M, et al. Retinoid effects on epidermal structure, differentiation, and permeability. Lab Invest. 1981;44:531-540.
- Williams ML, Elias PM. Nature of skin fragility in patients receiving retinoids for systemic effect. Arch Dermatol. 1981;117:611-619.
- Rubenstein R, Roenigk HH, Stegman SJ, et al. Atypical keloids after dermabrasion of patients taking isotretinoin. J Am Acad Dermatol. 1986;15:280-285. doi:10.1016/S0190-9622(86)70167-9
- Zachariae H. Delayed wound healing and keloid formation following argon laser treatment or dermabrasion during isotretinoin treatment. Br J Dermatol. 1988;118:703-706. doi:10.1111/j.1365-2133.1988.tb02574.x
- Katz BE, Mac Farlane DF. Atypical facial scarring after isotretinoin therapy in a patient with previous dermabrasion. J Am Acad Dermatol. 1994;30:852-853. doi:10.1016/S0190-9622(94)70096-6
Practice Points
- Isotretinoin is used to treat severe nodulocystic acne but can cause adverse effects such as skin fragility, xerosis, and poor wound healing.
- Dermatologists should inform athletes of heightened skin vulnerability while undergoing isotretinoin treatment.
- Isotretinoin-induced skin fragility involves the effects of isotretinoin on sebocytes, transepidermal water loss, and disruption of the integrity of the epidermis.
Study Finds Isotretinoin Effective for Acne in Transgender Patients on Hormone Rx
TOPLINE:
, but more information is needed on dosing and barriers to treatment.
METHODOLOGY:
- Acne can be a side effect of masculinizing hormone therapy for transmasculine individuals. While isotretinoin is an effective treatment option for acne, its effectiveness and safety in transgender and gender-diverse individuals are not well understood.
- This retrospective case series included 55 patients (mean age, 25.4 years) undergoing masculinizing hormone therapy at four medical centers, who were prescribed isotretinoin for acne associated with treatment.
- Isotretinoin treatment was started a median of 22.1 months after hormone therapy was initiated and continued for a median of 6 months with a median cumulative dose of 132.7 mg/kg.
- Researchers assessed acne improvement, clearance, recurrence, adverse effects, and reasons for treatment discontinuation.
TAKEAWAY:
- Overall, 48 patients (87.3%) experienced improvement, and 26 (47.3%) achieved clearance during treatment. A higher proportion of patients experienced improvement (97% vs 72.7%) and achieved clearance (63.6% vs 22.7%) with cumulative doses of ≥ 120 mg/kg than those who received cumulative doses < 120 mg/kg.
- The risk for recurrence was 20% (in four patients) among 20 patients who achieved clearance and had any subsequent health care encounters, with a mean follow-up time of 734.3 days.
- Common adverse effects included dryness (80%), joint pain (14.5%), and headaches (10.9%). Other adverse effects included nose bleeds (9.1%) and depression (5.5%).
- Of the 22 patients with a cumulative dose < 120 mg/kg, 14 (63.6%) were lost to follow-up; among those not lost to follow-up, 2 patients discontinued treatment because of transfer of care, 1 because of adverse effects, and 1 because of gender-affirming surgery, with concerns about wound healing.
IN PRACTICE:
“Although isotretinoin appears to be an effective treatment option for acne among individuals undergoing masculinizing hormone therapy, further efforts are needed to understand optimal dosing and treatment barriers to improve outcomes in transgender and gender-diverse individuals receiving testosterone,” the authors concluded.
SOURCE:
The study, led by James Choe, BS, Department of Dermatology, Brigham and Women’s Hospital, Harvard Medical School, Boston, was published online in JAMA Dermatology.
LIMITATIONS:
The study population was limited to four centers, and variability in clinician- and patient-reported acne outcomes and missing information could affect the reliability of data. Because of the small sample size, the association of masculinizing hormone therapy regimens with outcomes could not be evaluated.
DISCLOSURES:
One author is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Three authors reported receiving grants or personal fees from various sources. The other authors declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
, but more information is needed on dosing and barriers to treatment.
METHODOLOGY:
- Acne can be a side effect of masculinizing hormone therapy for transmasculine individuals. While isotretinoin is an effective treatment option for acne, its effectiveness and safety in transgender and gender-diverse individuals are not well understood.
- This retrospective case series included 55 patients (mean age, 25.4 years) undergoing masculinizing hormone therapy at four medical centers, who were prescribed isotretinoin for acne associated with treatment.
- Isotretinoin treatment was started a median of 22.1 months after hormone therapy was initiated and continued for a median of 6 months with a median cumulative dose of 132.7 mg/kg.
- Researchers assessed acne improvement, clearance, recurrence, adverse effects, and reasons for treatment discontinuation.
TAKEAWAY:
- Overall, 48 patients (87.3%) experienced improvement, and 26 (47.3%) achieved clearance during treatment. A higher proportion of patients experienced improvement (97% vs 72.7%) and achieved clearance (63.6% vs 22.7%) with cumulative doses of ≥ 120 mg/kg than those who received cumulative doses < 120 mg/kg.
- The risk for recurrence was 20% (in four patients) among 20 patients who achieved clearance and had any subsequent health care encounters, with a mean follow-up time of 734.3 days.
- Common adverse effects included dryness (80%), joint pain (14.5%), and headaches (10.9%). Other adverse effects included nose bleeds (9.1%) and depression (5.5%).
- Of the 22 patients with a cumulative dose < 120 mg/kg, 14 (63.6%) were lost to follow-up; among those not lost to follow-up, 2 patients discontinued treatment because of transfer of care, 1 because of adverse effects, and 1 because of gender-affirming surgery, with concerns about wound healing.
IN PRACTICE:
“Although isotretinoin appears to be an effective treatment option for acne among individuals undergoing masculinizing hormone therapy, further efforts are needed to understand optimal dosing and treatment barriers to improve outcomes in transgender and gender-diverse individuals receiving testosterone,” the authors concluded.
SOURCE:
The study, led by James Choe, BS, Department of Dermatology, Brigham and Women’s Hospital, Harvard Medical School, Boston, was published online in JAMA Dermatology.
LIMITATIONS:
The study population was limited to four centers, and variability in clinician- and patient-reported acne outcomes and missing information could affect the reliability of data. Because of the small sample size, the association of masculinizing hormone therapy regimens with outcomes could not be evaluated.
DISCLOSURES:
One author is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Three authors reported receiving grants or personal fees from various sources. The other authors declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
, but more information is needed on dosing and barriers to treatment.
METHODOLOGY:
- Acne can be a side effect of masculinizing hormone therapy for transmasculine individuals. While isotretinoin is an effective treatment option for acne, its effectiveness and safety in transgender and gender-diverse individuals are not well understood.
- This retrospective case series included 55 patients (mean age, 25.4 years) undergoing masculinizing hormone therapy at four medical centers, who were prescribed isotretinoin for acne associated with treatment.
- Isotretinoin treatment was started a median of 22.1 months after hormone therapy was initiated and continued for a median of 6 months with a median cumulative dose of 132.7 mg/kg.
- Researchers assessed acne improvement, clearance, recurrence, adverse effects, and reasons for treatment discontinuation.
TAKEAWAY:
- Overall, 48 patients (87.3%) experienced improvement, and 26 (47.3%) achieved clearance during treatment. A higher proportion of patients experienced improvement (97% vs 72.7%) and achieved clearance (63.6% vs 22.7%) with cumulative doses of ≥ 120 mg/kg than those who received cumulative doses < 120 mg/kg.
- The risk for recurrence was 20% (in four patients) among 20 patients who achieved clearance and had any subsequent health care encounters, with a mean follow-up time of 734.3 days.
- Common adverse effects included dryness (80%), joint pain (14.5%), and headaches (10.9%). Other adverse effects included nose bleeds (9.1%) and depression (5.5%).
- Of the 22 patients with a cumulative dose < 120 mg/kg, 14 (63.6%) were lost to follow-up; among those not lost to follow-up, 2 patients discontinued treatment because of transfer of care, 1 because of adverse effects, and 1 because of gender-affirming surgery, with concerns about wound healing.
IN PRACTICE:
“Although isotretinoin appears to be an effective treatment option for acne among individuals undergoing masculinizing hormone therapy, further efforts are needed to understand optimal dosing and treatment barriers to improve outcomes in transgender and gender-diverse individuals receiving testosterone,” the authors concluded.
SOURCE:
The study, led by James Choe, BS, Department of Dermatology, Brigham and Women’s Hospital, Harvard Medical School, Boston, was published online in JAMA Dermatology.
LIMITATIONS:
The study population was limited to four centers, and variability in clinician- and patient-reported acne outcomes and missing information could affect the reliability of data. Because of the small sample size, the association of masculinizing hormone therapy regimens with outcomes could not be evaluated.
DISCLOSURES:
One author is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Three authors reported receiving grants or personal fees from various sources. The other authors declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
Study Identifies Several Factors That Influence Longterm Antibiotic Prescribing for Acne
to follow them, according to the authors of a recently published study.
“This study explored why dermatologists still prescribe a good number of long-term antibiotics for people with acne,” the study’s senior author Howa Yeung, MD, MSc, assistant professor of dermatology at Emory University, Atlanta, said in an interview. “And we found a lot of reasons.” The study was published online in JAMA Dermatology.
Using online surveys and semi-structured video interviews of 30 dermatologists, infectious disease physicians with expertise in antimicrobial stewardship, dermatology residents, and nonphysician clinicians, the investigators assessed respondents’ knowledge and attitudes regarding long-term antibiotics in acne. Salient themes impacting long-term antibiotic prescriptions included the following:
- A perceived dearth of evidence to justify changes in practice.
- Difficulties with iPLEDGE, the Risk Evaluation and Mitigation Strategy (REMS) for managing the teratogenic risks associated with isotretinoin, and with discussing oral contraceptives.
- “Navigating” discussions with about tapering-off of antibiotics.
- Challenging patient demands.
- A lack of effective tools for monitoring progress in antibiotic stewardship.
“It’s surprising there are so many barriers that make it difficult for dermatologists to stick with the guidelines even if they want to,” said Dr. Yeung, a coauthor of the recently released updated American Academy of Dermatology (AAD) acne management guidelines.
A dermatologist who wants to stop systemic antibiotics within 3 months may not know how to do so, he explained, or high demand for appointments may prevent timely follow-ups.
A major reason why dermatologists struggle to limit long-term antibiotic use is that there are very few substitutes that are perceived to work as well, said David J. Margolis, MD, PhD, who was not involved with the study and was asked to comment on the results. He is professor of epidemiology and dermatology at the University of Pennsylvania, Philadelphia.
“Part of the reason antibiotics are being used to treat acne is that they’re effective, and effective for severe disease,” he said. The alternatives, which are mostly topicals, said Dr. Margolis, do not work as well for moderate to severe disease or, with isotretinoin, involve time-consuming hurdles. Dr. Margolis said that he often hears such concerns from individual dermatologists. “But it’s helpful to see these in a well-organized, well-reported qualitative study.”
Infectious disease specialists surveyed considered limiting long-term antibiotic use as extremely important, while several dermatologists “argued that other specialties ‘underestimate the impact acne has on people’s lives,’ ” the authors wrote. Other respondents prioritized making the right choice for the patient at hand.
Although guidelines were never meant to be black and white, Dr. Yeung said, it is crucial to target the goal of tapering off after about 3-4 months — a cutoff with which guidelines from groups including the AAD, the Japanese Dermatological Association in guidelines from 2016, and 2017, respectively, and others concur.
He added, “Some folks believe that if the oral antibiotic is working, why stop? We need to develop evidence to show that reducing oral antibiotic use is important to our patients, not just to a theoretical problem of antibiotic resistance in society.” For example, in a study published in The Lancet in 2004, patients who used strictly topical regimens achieved efficacy similar to that of those who used only oral antibiotics.
In addition, some clinicians worried that limiting antibiotics could reduce patient satisfaction, spurring switches to other providers. However, he and the other authors of the JAMA Dermatology study noted that in a survey of patients with acne published in the Journal of Clinical and Aesthetic Dermatology in 2019, 76.9% said they would be “very or extremely likely” to use effective antibiotic-free treatments if offered.
Because most respondents were highly aware of the importance of antibiotic stewardship, Dr. Yeung said, additional passive education is not necessarily the answer. “It will take a concerted effort by our national societies to come up with resources and solutions for individual dermatologists to overcome some of these larger barriers.” Such solutions could range from training in communication and shared decision-making to implementing systems that provide individualized feedback to support antibiotic stewardship.
Many ongoing studies are examining antibiotic stewardship, Dr. Margolis said in the interview. However, he added, dermatologists’ idea of long-term use is 3 months, versus 1 month or less in other specialties. “Moreover, dermatology patients tend to be much healthier individuals and are rarely hospitalized, so there may be some issues comparing the ongoing studies to individuals with acne.” Future research will need to account for such differences, he said.
The study was funded by an American Acne & Rosacea Society Clinical Research Award. Dr. Yeung is associate editor of JAMA Dermatology. Dr. Margolis has received a National Institutes of Health grant to study doxycycline versus spironolactone in acne.
to follow them, according to the authors of a recently published study.
“This study explored why dermatologists still prescribe a good number of long-term antibiotics for people with acne,” the study’s senior author Howa Yeung, MD, MSc, assistant professor of dermatology at Emory University, Atlanta, said in an interview. “And we found a lot of reasons.” The study was published online in JAMA Dermatology.
Using online surveys and semi-structured video interviews of 30 dermatologists, infectious disease physicians with expertise in antimicrobial stewardship, dermatology residents, and nonphysician clinicians, the investigators assessed respondents’ knowledge and attitudes regarding long-term antibiotics in acne. Salient themes impacting long-term antibiotic prescriptions included the following:
- A perceived dearth of evidence to justify changes in practice.
- Difficulties with iPLEDGE, the Risk Evaluation and Mitigation Strategy (REMS) for managing the teratogenic risks associated with isotretinoin, and with discussing oral contraceptives.
- “Navigating” discussions with about tapering-off of antibiotics.
- Challenging patient demands.
- A lack of effective tools for monitoring progress in antibiotic stewardship.
“It’s surprising there are so many barriers that make it difficult for dermatologists to stick with the guidelines even if they want to,” said Dr. Yeung, a coauthor of the recently released updated American Academy of Dermatology (AAD) acne management guidelines.
A dermatologist who wants to stop systemic antibiotics within 3 months may not know how to do so, he explained, or high demand for appointments may prevent timely follow-ups.
A major reason why dermatologists struggle to limit long-term antibiotic use is that there are very few substitutes that are perceived to work as well, said David J. Margolis, MD, PhD, who was not involved with the study and was asked to comment on the results. He is professor of epidemiology and dermatology at the University of Pennsylvania, Philadelphia.
“Part of the reason antibiotics are being used to treat acne is that they’re effective, and effective for severe disease,” he said. The alternatives, which are mostly topicals, said Dr. Margolis, do not work as well for moderate to severe disease or, with isotretinoin, involve time-consuming hurdles. Dr. Margolis said that he often hears such concerns from individual dermatologists. “But it’s helpful to see these in a well-organized, well-reported qualitative study.”
Infectious disease specialists surveyed considered limiting long-term antibiotic use as extremely important, while several dermatologists “argued that other specialties ‘underestimate the impact acne has on people’s lives,’ ” the authors wrote. Other respondents prioritized making the right choice for the patient at hand.
Although guidelines were never meant to be black and white, Dr. Yeung said, it is crucial to target the goal of tapering off after about 3-4 months — a cutoff with which guidelines from groups including the AAD, the Japanese Dermatological Association in guidelines from 2016, and 2017, respectively, and others concur.
He added, “Some folks believe that if the oral antibiotic is working, why stop? We need to develop evidence to show that reducing oral antibiotic use is important to our patients, not just to a theoretical problem of antibiotic resistance in society.” For example, in a study published in The Lancet in 2004, patients who used strictly topical regimens achieved efficacy similar to that of those who used only oral antibiotics.
In addition, some clinicians worried that limiting antibiotics could reduce patient satisfaction, spurring switches to other providers. However, he and the other authors of the JAMA Dermatology study noted that in a survey of patients with acne published in the Journal of Clinical and Aesthetic Dermatology in 2019, 76.9% said they would be “very or extremely likely” to use effective antibiotic-free treatments if offered.
Because most respondents were highly aware of the importance of antibiotic stewardship, Dr. Yeung said, additional passive education is not necessarily the answer. “It will take a concerted effort by our national societies to come up with resources and solutions for individual dermatologists to overcome some of these larger barriers.” Such solutions could range from training in communication and shared decision-making to implementing systems that provide individualized feedback to support antibiotic stewardship.
Many ongoing studies are examining antibiotic stewardship, Dr. Margolis said in the interview. However, he added, dermatologists’ idea of long-term use is 3 months, versus 1 month or less in other specialties. “Moreover, dermatology patients tend to be much healthier individuals and are rarely hospitalized, so there may be some issues comparing the ongoing studies to individuals with acne.” Future research will need to account for such differences, he said.
The study was funded by an American Acne & Rosacea Society Clinical Research Award. Dr. Yeung is associate editor of JAMA Dermatology. Dr. Margolis has received a National Institutes of Health grant to study doxycycline versus spironolactone in acne.
to follow them, according to the authors of a recently published study.
“This study explored why dermatologists still prescribe a good number of long-term antibiotics for people with acne,” the study’s senior author Howa Yeung, MD, MSc, assistant professor of dermatology at Emory University, Atlanta, said in an interview. “And we found a lot of reasons.” The study was published online in JAMA Dermatology.
Using online surveys and semi-structured video interviews of 30 dermatologists, infectious disease physicians with expertise in antimicrobial stewardship, dermatology residents, and nonphysician clinicians, the investigators assessed respondents’ knowledge and attitudes regarding long-term antibiotics in acne. Salient themes impacting long-term antibiotic prescriptions included the following:
- A perceived dearth of evidence to justify changes in practice.
- Difficulties with iPLEDGE, the Risk Evaluation and Mitigation Strategy (REMS) for managing the teratogenic risks associated with isotretinoin, and with discussing oral contraceptives.
- “Navigating” discussions with about tapering-off of antibiotics.
- Challenging patient demands.
- A lack of effective tools for monitoring progress in antibiotic stewardship.
“It’s surprising there are so many barriers that make it difficult for dermatologists to stick with the guidelines even if they want to,” said Dr. Yeung, a coauthor of the recently released updated American Academy of Dermatology (AAD) acne management guidelines.
A dermatologist who wants to stop systemic antibiotics within 3 months may not know how to do so, he explained, or high demand for appointments may prevent timely follow-ups.
A major reason why dermatologists struggle to limit long-term antibiotic use is that there are very few substitutes that are perceived to work as well, said David J. Margolis, MD, PhD, who was not involved with the study and was asked to comment on the results. He is professor of epidemiology and dermatology at the University of Pennsylvania, Philadelphia.
“Part of the reason antibiotics are being used to treat acne is that they’re effective, and effective for severe disease,” he said. The alternatives, which are mostly topicals, said Dr. Margolis, do not work as well for moderate to severe disease or, with isotretinoin, involve time-consuming hurdles. Dr. Margolis said that he often hears such concerns from individual dermatologists. “But it’s helpful to see these in a well-organized, well-reported qualitative study.”
Infectious disease specialists surveyed considered limiting long-term antibiotic use as extremely important, while several dermatologists “argued that other specialties ‘underestimate the impact acne has on people’s lives,’ ” the authors wrote. Other respondents prioritized making the right choice for the patient at hand.
Although guidelines were never meant to be black and white, Dr. Yeung said, it is crucial to target the goal of tapering off after about 3-4 months — a cutoff with which guidelines from groups including the AAD, the Japanese Dermatological Association in guidelines from 2016, and 2017, respectively, and others concur.
He added, “Some folks believe that if the oral antibiotic is working, why stop? We need to develop evidence to show that reducing oral antibiotic use is important to our patients, not just to a theoretical problem of antibiotic resistance in society.” For example, in a study published in The Lancet in 2004, patients who used strictly topical regimens achieved efficacy similar to that of those who used only oral antibiotics.
In addition, some clinicians worried that limiting antibiotics could reduce patient satisfaction, spurring switches to other providers. However, he and the other authors of the JAMA Dermatology study noted that in a survey of patients with acne published in the Journal of Clinical and Aesthetic Dermatology in 2019, 76.9% said they would be “very or extremely likely” to use effective antibiotic-free treatments if offered.
Because most respondents were highly aware of the importance of antibiotic stewardship, Dr. Yeung said, additional passive education is not necessarily the answer. “It will take a concerted effort by our national societies to come up with resources and solutions for individual dermatologists to overcome some of these larger barriers.” Such solutions could range from training in communication and shared decision-making to implementing systems that provide individualized feedback to support antibiotic stewardship.
Many ongoing studies are examining antibiotic stewardship, Dr. Margolis said in the interview. However, he added, dermatologists’ idea of long-term use is 3 months, versus 1 month or less in other specialties. “Moreover, dermatology patients tend to be much healthier individuals and are rarely hospitalized, so there may be some issues comparing the ongoing studies to individuals with acne.” Future research will need to account for such differences, he said.
The study was funded by an American Acne & Rosacea Society Clinical Research Award. Dr. Yeung is associate editor of JAMA Dermatology. Dr. Margolis has received a National Institutes of Health grant to study doxycycline versus spironolactone in acne.
FROM JAMA DERMATOLOGY
Benzene Detected in Benzoyl Peroxide Products: Debate On Implications Continues
Nine days after the independent laboratory, it published another report in Environmental Health Perspectives (EHP), on March 14, also warning about BP acne products.
The bottom line was the same: The laboratory, based in New Haven, Connecticut, said its analyses raise substantial concerns about the safety of BP-containing acne products currently on the market.
The lab’s results showed that the products can form over 800 times the conditionally restricted FDA concentration limit of 2 parts per million (ppm) of benzene, with both prescription and over-the-counter products affected.
“This is a problem of degradation, not contamination,” David Light, CEO and founder of Valisure, said in a telephone interview. BP can decompose into benzene, and exposure to benzene has been linked with a higher risk for leukemia and other blood cancers, according to the American Cancer Society.
In the wake of the findings, however, debate has erupted over the method and approach used by Valisure to test these products, with critics and companies contending that more “real-world” use data are needed. And the US Pharmacopeia (USP) is asking for full transparency about the testing methods.
In a March 8 statement, USP said the petition indicated that modified USP methods were used in the study, noting that “if changes are made to a USP method, complete validation data is necessary to demonstrate that a product meets USP standards.”
However, Valisure contended that drug products need to demonstrate stability over the entire life cycle, from shipment to continued use, emphasizing that constitutes the best “real-world” approach. It also defended the methodology it used.
The reports have led to a state of uncertainty about the use of BP products.
“Right now, we have more unknowns than anything else,” John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said in a video posted on X and YouTube, summarizing the findings released by Valisure on March 6 and 14. He was not involved in the Valisure research.
In a telephone interview, Dr. Barbieri said the report “needs to be taken seriously,” but he also believed the Valisure report is lacking information about testing under “real-world” conditions. He is calling for more information and more transparency about the data. What’s clear, Dr. Barbieri told this news organization, is that the findings about high benzene levels are not a manufacturing error. “It’s something to do with the molecule itself.”
Valisure’s Analyses
Valisure performed an initial analysis, using a method called gas chromatography-mass spectrometry, which is the FDA-preferred method for detecting benzene, Mr. Light said. It tested 175 acne products, 99 containing BP and 76 with other ingredients, such as salicylic acid. All the products without BP had no detectable benzene or values below 2 ppm, the FDA concentration limit for benzene.
Of the 99 BP products, 94 contained benzene without any elevated temperature incubation, according to Valisure. Using 50 °C (122 °F, the accepted pharmaceutical stability testing temperature) on 66 products, Valisure detected over 1500 ppm of benzene in two products, over 100 ppm in 17 products, and over 10 ppm in 42 products over an 18-day period.
The analysis confirmed, Valisure said in a press release and the petition, that a substantial amount of benzene can form in a BP product and leak outside the packaging into surrounding air.
The EHP paper, which includes authors from Valisure, reported that researchers took single lots of seven branded BP products, namely, Equate Beauty 2.5% BP cleansers, Neutrogena 10% BP cleanser, CVS Health 10% BP face wash, Walgreens 10% BP cream, Clean & Clear 10% cleanser, Equate Beauty 10% BP acne wash, and Proactiv 2.5% BP cleanser.
Using testing that involved gas chromatography-mass spectrometry, benzene was detected in all the BP products samples tested, and levels increased during incubation at body and shelf-life performance temperatures to more than 2 ppm. The authors concluded that the study “raises substantial concerns” about the safety of BP products currently on the market.
Methodology Debates
Two days after Valisure released its analysis on March 6, the USP reviewed the citizen’s petition filed by Valisure and called for more transparency around the testing methods.
“The petition referenced USP and indicated that modified USP methods and procedures were used in the study. The presence of unsafe levels of benzene should be taken seriously,” the statement said. The USP statement also noted that the Valisure analysis used modified USP methods and said that “if changes are made to a USP method, complete validation data is necessary to demonstrate that a product meets USP standards.”
In its statement, USP took issue with a practice known as accelerated thermal degradation, which it said Valisure used. USP said the approach involves raising the storage temperature of a product to higher than the temperature indicated on the label for the purpose of simulating degradation over a longer period. While the approach may be acceptable, USP said, the temperatures chosen may not be what is expected to happen to the products.
In response, Mr. Light of Valisure referenced guidance issued in August, 2020, from the FDA, stating that the method it used in the BP analysis can be used to detect impurities in hand sanitizers, including benzene. (In 2021, Valisure detected high levels of benzene in some hand sanitizers and asked the FDA to take action.)
Company Response
Among the companies that took issue with the report was Reckitt, which makes Clearasil, which contains BP. In a statement, the company said, in part: “The products and their ingredients are stable over the storage conditions described on their packaging which represent all reasonable and foreseeable conditions.” It said the findings presented by Valisure reflect “unrealistic scenarios rather than real-world conditions.”
The Personal Care Products Council, a national trade association that represents cosmetic and personal care product manufacturers, also took issue with the findings and the approach used to evaluate the products.
FDA and the Citizen’s Petition
The FDA accepted the petition, Mr. Light said, and gave it a docket number. “We’ll hopefully hear more soon” because the FDA is required to respond to a citizen’s petition within 180 days, he said.
“We generally don’t comment on pending citizens’ petitions,” an FDA spokesperson said in an email. “When we respond, we will respond directly to the petitioner and post the response in the designated agency public docket.”
Valisure’s Patent Application
Mr. Light and others have applied for a patent on methods of producing shelf-stable formulations to prevent degradation of BP to benzene.
“We saw the problem long before we had any sort of application,” Mr. Light said. The issue has been “known for decades.”
Role of BP Products for Acne
In the midst of uncertainty, “the first discussion is, do we want to use it?” Dr. Barbieri said in the interview. Some patients may want to avoid it altogether, until more data are available, including more verification of the findings, while others may be comfortable accepting the potential risk, he said.
“Benzoyl peroxide is one of our foundational acne treatments,” Dr. Barbieri said. In the American Academy of Dermatology updated guidelines on acne, published in January, 2024, strong recommendations were made for BP products, as well as topical retinoids, topical antibiotics, and oral doxycycline.
“When you take away BP, there’s no substitute for it,” Dr. Barbieri said. And if patients don’t get improvement with topicals, oral medications might be needed, and “these all have their own risks.”
In the Interim
Until more information is available, Dr. Barbieri is advising patients not to store the products at high temperatures or for a long time. Don’t keep the products past their expiration date, and perhaps keep products for a shorter time, “something like a month,” he said.
Those living in a hot climate might consider storing the products in the refrigerator, he said.
“We need more data from Valisure, from other groups that confirm their findings, and we need to hear from the FDA,” Dr. Barbieri said. “There’s a lot of uncertainty right now. But it’s important not to overreact.”
Dr. Barbieri had no relevant disclosures.
A version of this article appeared on Medscape.com.
Nine days after the independent laboratory, it published another report in Environmental Health Perspectives (EHP), on March 14, also warning about BP acne products.
The bottom line was the same: The laboratory, based in New Haven, Connecticut, said its analyses raise substantial concerns about the safety of BP-containing acne products currently on the market.
The lab’s results showed that the products can form over 800 times the conditionally restricted FDA concentration limit of 2 parts per million (ppm) of benzene, with both prescription and over-the-counter products affected.
“This is a problem of degradation, not contamination,” David Light, CEO and founder of Valisure, said in a telephone interview. BP can decompose into benzene, and exposure to benzene has been linked with a higher risk for leukemia and other blood cancers, according to the American Cancer Society.
In the wake of the findings, however, debate has erupted over the method and approach used by Valisure to test these products, with critics and companies contending that more “real-world” use data are needed. And the US Pharmacopeia (USP) is asking for full transparency about the testing methods.
In a March 8 statement, USP said the petition indicated that modified USP methods were used in the study, noting that “if changes are made to a USP method, complete validation data is necessary to demonstrate that a product meets USP standards.”
However, Valisure contended that drug products need to demonstrate stability over the entire life cycle, from shipment to continued use, emphasizing that constitutes the best “real-world” approach. It also defended the methodology it used.
The reports have led to a state of uncertainty about the use of BP products.
“Right now, we have more unknowns than anything else,” John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said in a video posted on X and YouTube, summarizing the findings released by Valisure on March 6 and 14. He was not involved in the Valisure research.
In a telephone interview, Dr. Barbieri said the report “needs to be taken seriously,” but he also believed the Valisure report is lacking information about testing under “real-world” conditions. He is calling for more information and more transparency about the data. What’s clear, Dr. Barbieri told this news organization, is that the findings about high benzene levels are not a manufacturing error. “It’s something to do with the molecule itself.”
Valisure’s Analyses
Valisure performed an initial analysis, using a method called gas chromatography-mass spectrometry, which is the FDA-preferred method for detecting benzene, Mr. Light said. It tested 175 acne products, 99 containing BP and 76 with other ingredients, such as salicylic acid. All the products without BP had no detectable benzene or values below 2 ppm, the FDA concentration limit for benzene.
Of the 99 BP products, 94 contained benzene without any elevated temperature incubation, according to Valisure. Using 50 °C (122 °F, the accepted pharmaceutical stability testing temperature) on 66 products, Valisure detected over 1500 ppm of benzene in two products, over 100 ppm in 17 products, and over 10 ppm in 42 products over an 18-day period.
The analysis confirmed, Valisure said in a press release and the petition, that a substantial amount of benzene can form in a BP product and leak outside the packaging into surrounding air.
The EHP paper, which includes authors from Valisure, reported that researchers took single lots of seven branded BP products, namely, Equate Beauty 2.5% BP cleansers, Neutrogena 10% BP cleanser, CVS Health 10% BP face wash, Walgreens 10% BP cream, Clean & Clear 10% cleanser, Equate Beauty 10% BP acne wash, and Proactiv 2.5% BP cleanser.
Using testing that involved gas chromatography-mass spectrometry, benzene was detected in all the BP products samples tested, and levels increased during incubation at body and shelf-life performance temperatures to more than 2 ppm. The authors concluded that the study “raises substantial concerns” about the safety of BP products currently on the market.
Methodology Debates
Two days after Valisure released its analysis on March 6, the USP reviewed the citizen’s petition filed by Valisure and called for more transparency around the testing methods.
“The petition referenced USP and indicated that modified USP methods and procedures were used in the study. The presence of unsafe levels of benzene should be taken seriously,” the statement said. The USP statement also noted that the Valisure analysis used modified USP methods and said that “if changes are made to a USP method, complete validation data is necessary to demonstrate that a product meets USP standards.”
In its statement, USP took issue with a practice known as accelerated thermal degradation, which it said Valisure used. USP said the approach involves raising the storage temperature of a product to higher than the temperature indicated on the label for the purpose of simulating degradation over a longer period. While the approach may be acceptable, USP said, the temperatures chosen may not be what is expected to happen to the products.
In response, Mr. Light of Valisure referenced guidance issued in August, 2020, from the FDA, stating that the method it used in the BP analysis can be used to detect impurities in hand sanitizers, including benzene. (In 2021, Valisure detected high levels of benzene in some hand sanitizers and asked the FDA to take action.)
Company Response
Among the companies that took issue with the report was Reckitt, which makes Clearasil, which contains BP. In a statement, the company said, in part: “The products and their ingredients are stable over the storage conditions described on their packaging which represent all reasonable and foreseeable conditions.” It said the findings presented by Valisure reflect “unrealistic scenarios rather than real-world conditions.”
The Personal Care Products Council, a national trade association that represents cosmetic and personal care product manufacturers, also took issue with the findings and the approach used to evaluate the products.
FDA and the Citizen’s Petition
The FDA accepted the petition, Mr. Light said, and gave it a docket number. “We’ll hopefully hear more soon” because the FDA is required to respond to a citizen’s petition within 180 days, he said.
“We generally don’t comment on pending citizens’ petitions,” an FDA spokesperson said in an email. “When we respond, we will respond directly to the petitioner and post the response in the designated agency public docket.”
Valisure’s Patent Application
Mr. Light and others have applied for a patent on methods of producing shelf-stable formulations to prevent degradation of BP to benzene.
“We saw the problem long before we had any sort of application,” Mr. Light said. The issue has been “known for decades.”
Role of BP Products for Acne
In the midst of uncertainty, “the first discussion is, do we want to use it?” Dr. Barbieri said in the interview. Some patients may want to avoid it altogether, until more data are available, including more verification of the findings, while others may be comfortable accepting the potential risk, he said.
“Benzoyl peroxide is one of our foundational acne treatments,” Dr. Barbieri said. In the American Academy of Dermatology updated guidelines on acne, published in January, 2024, strong recommendations were made for BP products, as well as topical retinoids, topical antibiotics, and oral doxycycline.
“When you take away BP, there’s no substitute for it,” Dr. Barbieri said. And if patients don’t get improvement with topicals, oral medications might be needed, and “these all have their own risks.”
In the Interim
Until more information is available, Dr. Barbieri is advising patients not to store the products at high temperatures or for a long time. Don’t keep the products past their expiration date, and perhaps keep products for a shorter time, “something like a month,” he said.
Those living in a hot climate might consider storing the products in the refrigerator, he said.
“We need more data from Valisure, from other groups that confirm their findings, and we need to hear from the FDA,” Dr. Barbieri said. “There’s a lot of uncertainty right now. But it’s important not to overreact.”
Dr. Barbieri had no relevant disclosures.
A version of this article appeared on Medscape.com.
Nine days after the independent laboratory, it published another report in Environmental Health Perspectives (EHP), on March 14, also warning about BP acne products.
The bottom line was the same: The laboratory, based in New Haven, Connecticut, said its analyses raise substantial concerns about the safety of BP-containing acne products currently on the market.
The lab’s results showed that the products can form over 800 times the conditionally restricted FDA concentration limit of 2 parts per million (ppm) of benzene, with both prescription and over-the-counter products affected.
“This is a problem of degradation, not contamination,” David Light, CEO and founder of Valisure, said in a telephone interview. BP can decompose into benzene, and exposure to benzene has been linked with a higher risk for leukemia and other blood cancers, according to the American Cancer Society.
In the wake of the findings, however, debate has erupted over the method and approach used by Valisure to test these products, with critics and companies contending that more “real-world” use data are needed. And the US Pharmacopeia (USP) is asking for full transparency about the testing methods.
In a March 8 statement, USP said the petition indicated that modified USP methods were used in the study, noting that “if changes are made to a USP method, complete validation data is necessary to demonstrate that a product meets USP standards.”
However, Valisure contended that drug products need to demonstrate stability over the entire life cycle, from shipment to continued use, emphasizing that constitutes the best “real-world” approach. It also defended the methodology it used.
The reports have led to a state of uncertainty about the use of BP products.
“Right now, we have more unknowns than anything else,” John Barbieri, MD, MBA, assistant professor of dermatology at Harvard Medical School and director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, said in a video posted on X and YouTube, summarizing the findings released by Valisure on March 6 and 14. He was not involved in the Valisure research.
In a telephone interview, Dr. Barbieri said the report “needs to be taken seriously,” but he also believed the Valisure report is lacking information about testing under “real-world” conditions. He is calling for more information and more transparency about the data. What’s clear, Dr. Barbieri told this news organization, is that the findings about high benzene levels are not a manufacturing error. “It’s something to do with the molecule itself.”
Valisure’s Analyses
Valisure performed an initial analysis, using a method called gas chromatography-mass spectrometry, which is the FDA-preferred method for detecting benzene, Mr. Light said. It tested 175 acne products, 99 containing BP and 76 with other ingredients, such as salicylic acid. All the products without BP had no detectable benzene or values below 2 ppm, the FDA concentration limit for benzene.
Of the 99 BP products, 94 contained benzene without any elevated temperature incubation, according to Valisure. Using 50 °C (122 °F, the accepted pharmaceutical stability testing temperature) on 66 products, Valisure detected over 1500 ppm of benzene in two products, over 100 ppm in 17 products, and over 10 ppm in 42 products over an 18-day period.
The analysis confirmed, Valisure said in a press release and the petition, that a substantial amount of benzene can form in a BP product and leak outside the packaging into surrounding air.
The EHP paper, which includes authors from Valisure, reported that researchers took single lots of seven branded BP products, namely, Equate Beauty 2.5% BP cleansers, Neutrogena 10% BP cleanser, CVS Health 10% BP face wash, Walgreens 10% BP cream, Clean & Clear 10% cleanser, Equate Beauty 10% BP acne wash, and Proactiv 2.5% BP cleanser.
Using testing that involved gas chromatography-mass spectrometry, benzene was detected in all the BP products samples tested, and levels increased during incubation at body and shelf-life performance temperatures to more than 2 ppm. The authors concluded that the study “raises substantial concerns” about the safety of BP products currently on the market.
Methodology Debates
Two days after Valisure released its analysis on March 6, the USP reviewed the citizen’s petition filed by Valisure and called for more transparency around the testing methods.
“The petition referenced USP and indicated that modified USP methods and procedures were used in the study. The presence of unsafe levels of benzene should be taken seriously,” the statement said. The USP statement also noted that the Valisure analysis used modified USP methods and said that “if changes are made to a USP method, complete validation data is necessary to demonstrate that a product meets USP standards.”
In its statement, USP took issue with a practice known as accelerated thermal degradation, which it said Valisure used. USP said the approach involves raising the storage temperature of a product to higher than the temperature indicated on the label for the purpose of simulating degradation over a longer period. While the approach may be acceptable, USP said, the temperatures chosen may not be what is expected to happen to the products.
In response, Mr. Light of Valisure referenced guidance issued in August, 2020, from the FDA, stating that the method it used in the BP analysis can be used to detect impurities in hand sanitizers, including benzene. (In 2021, Valisure detected high levels of benzene in some hand sanitizers and asked the FDA to take action.)
Company Response
Among the companies that took issue with the report was Reckitt, which makes Clearasil, which contains BP. In a statement, the company said, in part: “The products and their ingredients are stable over the storage conditions described on their packaging which represent all reasonable and foreseeable conditions.” It said the findings presented by Valisure reflect “unrealistic scenarios rather than real-world conditions.”
The Personal Care Products Council, a national trade association that represents cosmetic and personal care product manufacturers, also took issue with the findings and the approach used to evaluate the products.
FDA and the Citizen’s Petition
The FDA accepted the petition, Mr. Light said, and gave it a docket number. “We’ll hopefully hear more soon” because the FDA is required to respond to a citizen’s petition within 180 days, he said.
“We generally don’t comment on pending citizens’ petitions,” an FDA spokesperson said in an email. “When we respond, we will respond directly to the petitioner and post the response in the designated agency public docket.”
Valisure’s Patent Application
Mr. Light and others have applied for a patent on methods of producing shelf-stable formulations to prevent degradation of BP to benzene.
“We saw the problem long before we had any sort of application,” Mr. Light said. The issue has been “known for decades.”
Role of BP Products for Acne
In the midst of uncertainty, “the first discussion is, do we want to use it?” Dr. Barbieri said in the interview. Some patients may want to avoid it altogether, until more data are available, including more verification of the findings, while others may be comfortable accepting the potential risk, he said.
“Benzoyl peroxide is one of our foundational acne treatments,” Dr. Barbieri said. In the American Academy of Dermatology updated guidelines on acne, published in January, 2024, strong recommendations were made for BP products, as well as topical retinoids, topical antibiotics, and oral doxycycline.
“When you take away BP, there’s no substitute for it,” Dr. Barbieri said. And if patients don’t get improvement with topicals, oral medications might be needed, and “these all have their own risks.”
In the Interim
Until more information is available, Dr. Barbieri is advising patients not to store the products at high temperatures or for a long time. Don’t keep the products past their expiration date, and perhaps keep products for a shorter time, “something like a month,” he said.
Those living in a hot climate might consider storing the products in the refrigerator, he said.
“We need more data from Valisure, from other groups that confirm their findings, and we need to hear from the FDA,” Dr. Barbieri said. “There’s a lot of uncertainty right now. But it’s important not to overreact.”
Dr. Barbieri had no relevant disclosures.
A version of this article appeared on Medscape.com.
Acne Risk With Progestin-Only Long-Acting Reversible Contraceptives Evaluated
TOPLINE:
Despite the .
METHODOLOGY:
- Progestin-only LARC may increase the risk for acne, but this has not been well studied in adolescents and young adults.
- In the study, researchers evaluated the incidence of acne, acne as a reason for removal, and strategies used to manage acne after insertion of a progestin-only intrauterine device (IUD) or contraceptive implant in 1319 adolescents and young adults across four Adolescent Medicine LARC Collaborative study sites from January 2017 to June 2021.The mean age at insertion was 18.6 years.
- Overall, 24% of participants had acne at the time of LARC insertion.
- Worsening acne was defined as new patient reports of concern about acne, observations of acne, or addition of an acne medication after insertion; increased severity noted on an exam during follow-up or at the time of LARC removal; or acne reported as a side effect and/or reason for LARC removal.
TAKEAWAY:
- During the study period, 376 participants (28.5%) experienced worsening acne after LARC insertion, and 17% reported acne as a new concern, with no differences between those who received an IUD or an implant.
- Only 44 of the 376 participants (11.7%) who reported worsening acne were being treated with an oral agent at follow-up.
- Of the 542 individuals (41% of the total) who had the LARC device removed, 40 (7.4%) cited concerns about acne for removing the device, although just 5 (0.92%) said that acne was the only reason for removal. Of the 40 with concerns about acne when the device was removed, 18 (45%) had documented acne at the time of insertion.
IN PRACTICE:
The authors recommend that clinicians prescribing progestin-only LARC should counsel patients that acne may be a side effect, reassuring them that if they develop acne, “it typically is not problematic enough to warrant discontinuation,” and concluded that “concerns about the development or worsening of acne should not be cause to avoid these forms of contraception.”
SOURCE:
The study, led by Markus D. Boos, MD, PhD, of the division of dermatology in the Department of Pediatrics, University of Washington in Seattle and Seattle Children’s Hospital, was published in Pediatric Dermatology.
LIMITATIONS:
Individuals without documented acne were assumed to be acne-free, creating potential bias. Acne evaluation and treatment were not standardized and were not performed by dermatologists; acne severity was not recorded for many participants, possibly underestimating severity, and excluding LARC insertions without follow-up or with removal within 8 weeks may have underestimated the percentage of participants who developed new or worsening acne.
DISCLOSURES:
The study was supported by Investigator-Initiated Studies Program of Organon and by the Health Resources and Services Administration of the US Department of Health and Human Services. Many authors received grants for this work. The authors did not disclose any other competing interests.
A version of this article appeared on Medscape.com.
TOPLINE:
Despite the .
METHODOLOGY:
- Progestin-only LARC may increase the risk for acne, but this has not been well studied in adolescents and young adults.
- In the study, researchers evaluated the incidence of acne, acne as a reason for removal, and strategies used to manage acne after insertion of a progestin-only intrauterine device (IUD) or contraceptive implant in 1319 adolescents and young adults across four Adolescent Medicine LARC Collaborative study sites from January 2017 to June 2021.The mean age at insertion was 18.6 years.
- Overall, 24% of participants had acne at the time of LARC insertion.
- Worsening acne was defined as new patient reports of concern about acne, observations of acne, or addition of an acne medication after insertion; increased severity noted on an exam during follow-up or at the time of LARC removal; or acne reported as a side effect and/or reason for LARC removal.
TAKEAWAY:
- During the study period, 376 participants (28.5%) experienced worsening acne after LARC insertion, and 17% reported acne as a new concern, with no differences between those who received an IUD or an implant.
- Only 44 of the 376 participants (11.7%) who reported worsening acne were being treated with an oral agent at follow-up.
- Of the 542 individuals (41% of the total) who had the LARC device removed, 40 (7.4%) cited concerns about acne for removing the device, although just 5 (0.92%) said that acne was the only reason for removal. Of the 40 with concerns about acne when the device was removed, 18 (45%) had documented acne at the time of insertion.
IN PRACTICE:
The authors recommend that clinicians prescribing progestin-only LARC should counsel patients that acne may be a side effect, reassuring them that if they develop acne, “it typically is not problematic enough to warrant discontinuation,” and concluded that “concerns about the development or worsening of acne should not be cause to avoid these forms of contraception.”
SOURCE:
The study, led by Markus D. Boos, MD, PhD, of the division of dermatology in the Department of Pediatrics, University of Washington in Seattle and Seattle Children’s Hospital, was published in Pediatric Dermatology.
LIMITATIONS:
Individuals without documented acne were assumed to be acne-free, creating potential bias. Acne evaluation and treatment were not standardized and were not performed by dermatologists; acne severity was not recorded for many participants, possibly underestimating severity, and excluding LARC insertions without follow-up or with removal within 8 weeks may have underestimated the percentage of participants who developed new or worsening acne.
DISCLOSURES:
The study was supported by Investigator-Initiated Studies Program of Organon and by the Health Resources and Services Administration of the US Department of Health and Human Services. Many authors received grants for this work. The authors did not disclose any other competing interests.
A version of this article appeared on Medscape.com.
TOPLINE:
Despite the .
METHODOLOGY:
- Progestin-only LARC may increase the risk for acne, but this has not been well studied in adolescents and young adults.
- In the study, researchers evaluated the incidence of acne, acne as a reason for removal, and strategies used to manage acne after insertion of a progestin-only intrauterine device (IUD) or contraceptive implant in 1319 adolescents and young adults across four Adolescent Medicine LARC Collaborative study sites from January 2017 to June 2021.The mean age at insertion was 18.6 years.
- Overall, 24% of participants had acne at the time of LARC insertion.
- Worsening acne was defined as new patient reports of concern about acne, observations of acne, or addition of an acne medication after insertion; increased severity noted on an exam during follow-up or at the time of LARC removal; or acne reported as a side effect and/or reason for LARC removal.
TAKEAWAY:
- During the study period, 376 participants (28.5%) experienced worsening acne after LARC insertion, and 17% reported acne as a new concern, with no differences between those who received an IUD or an implant.
- Only 44 of the 376 participants (11.7%) who reported worsening acne were being treated with an oral agent at follow-up.
- Of the 542 individuals (41% of the total) who had the LARC device removed, 40 (7.4%) cited concerns about acne for removing the device, although just 5 (0.92%) said that acne was the only reason for removal. Of the 40 with concerns about acne when the device was removed, 18 (45%) had documented acne at the time of insertion.
IN PRACTICE:
The authors recommend that clinicians prescribing progestin-only LARC should counsel patients that acne may be a side effect, reassuring them that if they develop acne, “it typically is not problematic enough to warrant discontinuation,” and concluded that “concerns about the development or worsening of acne should not be cause to avoid these forms of contraception.”
SOURCE:
The study, led by Markus D. Boos, MD, PhD, of the division of dermatology in the Department of Pediatrics, University of Washington in Seattle and Seattle Children’s Hospital, was published in Pediatric Dermatology.
LIMITATIONS:
Individuals without documented acne were assumed to be acne-free, creating potential bias. Acne evaluation and treatment were not standardized and were not performed by dermatologists; acne severity was not recorded for many participants, possibly underestimating severity, and excluding LARC insertions without follow-up or with removal within 8 weeks may have underestimated the percentage of participants who developed new or worsening acne.
DISCLOSURES:
The study was supported by Investigator-Initiated Studies Program of Organon and by the Health Resources and Services Administration of the US Department of Health and Human Services. Many authors received grants for this work. The authors did not disclose any other competing interests.
A version of this article appeared on Medscape.com.
Acne in Transmasculine Patients: Management Recommendations
SAN DIEGO — , a dermatologist told colleagues in a session at the American Academy of Dermatology annual meeting.
In these patients, treatment of acne is crucial, said Howa Yeung, MD, MSc, assistant professor of dermatology, Emory University, Atlanta. “These are patients who are suffering and reporting that they’re having mental health impacts” related to acne.
In transmasculine patients — those who were biologically female at birth but identify as masculine — testosterone therapy greatly boosts the risk for acne, even in adults who are long past adolescence, Dr. Yeung said. Data suggest that acne appears within the first 6 months after testosterone therapy begins, he said, “and the maximal and complete effect occurs within 1-2 years.”
A 2021 study tracked 988 transgender patients receiving testosterone at Fenway Health in Boston and found that 31% had a diagnosis of acne, up from 6.3% prior to taking hormones. And 2 years following the start of therapy, 25.1% had acne, with cases especially common among those aged 18-20.75 years (29.6%). Even among those aged 28.25-66.5 years, 17.1% had acne.
Transmasculine patients may develop acne in areas across the body “in places that you normally won’t see by just looking at the patient,” Dr. Yeung said. Excoriation in addition to comedones, papules, pustules, and nodules can be common, he added.
Dr. Yeung highlighted a 2019 study of transgender men that linked higher levels of acne to higher levels of serum testosterone, higher body mass index, and current smoking. And in a 2014 study, 6% of 50 transmasculine patients had moderate to severe acne after an average of 10 years on testosterone therapy.
A 2020 study of 696 transgender adults surveyed in California and Georgia found that 14% of transmasculine patients had moderate to severe acne — two thirds attributed it to hormone therapy — vs 1% of transfeminine patients, said Dr. Yeung, the lead author of the study. However, transmasculine patients were less likely to have seen a dermatologist.
Dr. Yeung also highlighted a 2021 study he coauthored that linked current moderate to severe acne in transmasculine patients taking testosterone to higher levels of depression and anxiety vs counterparts who had never had those forms of acne.
Another factor affecting acne in transmasculine patients is the use of chest binders to reduce breast size. “Wearing a chest binder is really helpful for a lot of our patients and is associated with improved self-esteem, mood, mental health, and safety in public,” Dr. Yeung said. However, the binders can contribute to skin problems.
Dr. Yeung said he and his colleagues emphasize the importance of breathable material in binders and suggest to patients that they not wear them when they’re in “safe spaces.”
Isotretinoin, Contraception Considerations
As for treatment of acne in transgender patients, Dr. Yeung cautioned colleagues to not automatically reject isotretinoin as an option for transgender patients who have a history of depression. Dermatologists may be tempted to avoid the drug in these patients because of its link to suicide, he said. (This apparent association has long been debated.) But, Dr. Yeung said, it’s important to consider that many of these patients suffered from anxiety and depression because of the lack of access to proper gender-reassignment treatment.
When using isotretinoin, he emphasized, it’s crucial to consider whether transmasculine patients could become pregnant while on this therapy. Consider whether the patient has the organs needed to become pregnant and ask questions about the potential that they could be impregnated.
“Remember that sexual behavior is different from gender identity,” Dr. Yeung said. A transmasculine person with a uterus and vagina, for example, may still have vaginal intercourse with males and potentially become pregnant. “So, we need to assess what kind of sexual behavior our patients are taking part in.”
Contraceptives such as intrauterine devices, implants, and injectable options may be helpful for transmasculine patients because they can reduce menstrual symptoms like spotting that can be distressing to them, he said. By helping a patient take a contraceptive, “you may actually be helping with their gender dysphoria and helping them get on isotretinoin.”
Dr. Yeung disclosed fees from JAMA and American Academy of Dermatology; grants/research funding from the American Acne & Rosacea Society, Dermatology Foundation, Department of Veterans Affairs, National Eczema Association, and National Institutes of Health; and speaker/faculty education honoraria from Dermatology Digest.
A version of this article appeared on Medscape.com.
SAN DIEGO — , a dermatologist told colleagues in a session at the American Academy of Dermatology annual meeting.
In these patients, treatment of acne is crucial, said Howa Yeung, MD, MSc, assistant professor of dermatology, Emory University, Atlanta. “These are patients who are suffering and reporting that they’re having mental health impacts” related to acne.
In transmasculine patients — those who were biologically female at birth but identify as masculine — testosterone therapy greatly boosts the risk for acne, even in adults who are long past adolescence, Dr. Yeung said. Data suggest that acne appears within the first 6 months after testosterone therapy begins, he said, “and the maximal and complete effect occurs within 1-2 years.”
A 2021 study tracked 988 transgender patients receiving testosterone at Fenway Health in Boston and found that 31% had a diagnosis of acne, up from 6.3% prior to taking hormones. And 2 years following the start of therapy, 25.1% had acne, with cases especially common among those aged 18-20.75 years (29.6%). Even among those aged 28.25-66.5 years, 17.1% had acne.
Transmasculine patients may develop acne in areas across the body “in places that you normally won’t see by just looking at the patient,” Dr. Yeung said. Excoriation in addition to comedones, papules, pustules, and nodules can be common, he added.
Dr. Yeung highlighted a 2019 study of transgender men that linked higher levels of acne to higher levels of serum testosterone, higher body mass index, and current smoking. And in a 2014 study, 6% of 50 transmasculine patients had moderate to severe acne after an average of 10 years on testosterone therapy.
A 2020 study of 696 transgender adults surveyed in California and Georgia found that 14% of transmasculine patients had moderate to severe acne — two thirds attributed it to hormone therapy — vs 1% of transfeminine patients, said Dr. Yeung, the lead author of the study. However, transmasculine patients were less likely to have seen a dermatologist.
Dr. Yeung also highlighted a 2021 study he coauthored that linked current moderate to severe acne in transmasculine patients taking testosterone to higher levels of depression and anxiety vs counterparts who had never had those forms of acne.
Another factor affecting acne in transmasculine patients is the use of chest binders to reduce breast size. “Wearing a chest binder is really helpful for a lot of our patients and is associated with improved self-esteem, mood, mental health, and safety in public,” Dr. Yeung said. However, the binders can contribute to skin problems.
Dr. Yeung said he and his colleagues emphasize the importance of breathable material in binders and suggest to patients that they not wear them when they’re in “safe spaces.”
Isotretinoin, Contraception Considerations
As for treatment of acne in transgender patients, Dr. Yeung cautioned colleagues to not automatically reject isotretinoin as an option for transgender patients who have a history of depression. Dermatologists may be tempted to avoid the drug in these patients because of its link to suicide, he said. (This apparent association has long been debated.) But, Dr. Yeung said, it’s important to consider that many of these patients suffered from anxiety and depression because of the lack of access to proper gender-reassignment treatment.
When using isotretinoin, he emphasized, it’s crucial to consider whether transmasculine patients could become pregnant while on this therapy. Consider whether the patient has the organs needed to become pregnant and ask questions about the potential that they could be impregnated.
“Remember that sexual behavior is different from gender identity,” Dr. Yeung said. A transmasculine person with a uterus and vagina, for example, may still have vaginal intercourse with males and potentially become pregnant. “So, we need to assess what kind of sexual behavior our patients are taking part in.”
Contraceptives such as intrauterine devices, implants, and injectable options may be helpful for transmasculine patients because they can reduce menstrual symptoms like spotting that can be distressing to them, he said. By helping a patient take a contraceptive, “you may actually be helping with their gender dysphoria and helping them get on isotretinoin.”
Dr. Yeung disclosed fees from JAMA and American Academy of Dermatology; grants/research funding from the American Acne & Rosacea Society, Dermatology Foundation, Department of Veterans Affairs, National Eczema Association, and National Institutes of Health; and speaker/faculty education honoraria from Dermatology Digest.
A version of this article appeared on Medscape.com.
SAN DIEGO — , a dermatologist told colleagues in a session at the American Academy of Dermatology annual meeting.
In these patients, treatment of acne is crucial, said Howa Yeung, MD, MSc, assistant professor of dermatology, Emory University, Atlanta. “These are patients who are suffering and reporting that they’re having mental health impacts” related to acne.
In transmasculine patients — those who were biologically female at birth but identify as masculine — testosterone therapy greatly boosts the risk for acne, even in adults who are long past adolescence, Dr. Yeung said. Data suggest that acne appears within the first 6 months after testosterone therapy begins, he said, “and the maximal and complete effect occurs within 1-2 years.”
A 2021 study tracked 988 transgender patients receiving testosterone at Fenway Health in Boston and found that 31% had a diagnosis of acne, up from 6.3% prior to taking hormones. And 2 years following the start of therapy, 25.1% had acne, with cases especially common among those aged 18-20.75 years (29.6%). Even among those aged 28.25-66.5 years, 17.1% had acne.
Transmasculine patients may develop acne in areas across the body “in places that you normally won’t see by just looking at the patient,” Dr. Yeung said. Excoriation in addition to comedones, papules, pustules, and nodules can be common, he added.
Dr. Yeung highlighted a 2019 study of transgender men that linked higher levels of acne to higher levels of serum testosterone, higher body mass index, and current smoking. And in a 2014 study, 6% of 50 transmasculine patients had moderate to severe acne after an average of 10 years on testosterone therapy.
A 2020 study of 696 transgender adults surveyed in California and Georgia found that 14% of transmasculine patients had moderate to severe acne — two thirds attributed it to hormone therapy — vs 1% of transfeminine patients, said Dr. Yeung, the lead author of the study. However, transmasculine patients were less likely to have seen a dermatologist.
Dr. Yeung also highlighted a 2021 study he coauthored that linked current moderate to severe acne in transmasculine patients taking testosterone to higher levels of depression and anxiety vs counterparts who had never had those forms of acne.
Another factor affecting acne in transmasculine patients is the use of chest binders to reduce breast size. “Wearing a chest binder is really helpful for a lot of our patients and is associated with improved self-esteem, mood, mental health, and safety in public,” Dr. Yeung said. However, the binders can contribute to skin problems.
Dr. Yeung said he and his colleagues emphasize the importance of breathable material in binders and suggest to patients that they not wear them when they’re in “safe spaces.”
Isotretinoin, Contraception Considerations
As for treatment of acne in transgender patients, Dr. Yeung cautioned colleagues to not automatically reject isotretinoin as an option for transgender patients who have a history of depression. Dermatologists may be tempted to avoid the drug in these patients because of its link to suicide, he said. (This apparent association has long been debated.) But, Dr. Yeung said, it’s important to consider that many of these patients suffered from anxiety and depression because of the lack of access to proper gender-reassignment treatment.
When using isotretinoin, he emphasized, it’s crucial to consider whether transmasculine patients could become pregnant while on this therapy. Consider whether the patient has the organs needed to become pregnant and ask questions about the potential that they could be impregnated.
“Remember that sexual behavior is different from gender identity,” Dr. Yeung said. A transmasculine person with a uterus and vagina, for example, may still have vaginal intercourse with males and potentially become pregnant. “So, we need to assess what kind of sexual behavior our patients are taking part in.”
Contraceptives such as intrauterine devices, implants, and injectable options may be helpful for transmasculine patients because they can reduce menstrual symptoms like spotting that can be distressing to them, he said. By helping a patient take a contraceptive, “you may actually be helping with their gender dysphoria and helping them get on isotretinoin.”
Dr. Yeung disclosed fees from JAMA and American Academy of Dermatology; grants/research funding from the American Acne & Rosacea Society, Dermatology Foundation, Department of Veterans Affairs, National Eczema Association, and National Institutes of Health; and speaker/faculty education honoraria from Dermatology Digest.
A version of this article appeared on Medscape.com.
FROM AAD 2024